A literature review: pre-/post-operative atrial fibrillation for thoracic aortic aneurysm procedures
Abstract
Atrial fibrillation (AF) is among the most frequent cardiac surgical arrhythmias documented. The global AF prevalence is estimated at over 33 million cases, with estimates ranging up to 6.1 million cases in the United States. Among cardiac surgical patients, the risk factors for new-onset post-operative AF (POAF) include Caucasian race with increased prevalence documented in older men. Due to trends of earlier thoracic aortic aneurysm (TAA) detection and treatment, it is timely to review the AF association with adverse TAA clinical outcomes. Towards this goal, a comprehensive PubMed literature review was performed. For this initial Medline literature search, the MeSH search strategy included “thoracic aortic aneurysm” and “atrial fibrillation”. Based on the pertinent articles identified, the limited literature available for preoperative TAA AF and the predictors of POAF following TAA procedures were reviewed. Given only a handful of publications addressing these pre-/post-operative AF topics were identified using this very broad initial search approach, a knowledge chasm exists–as very little is known about TAA patients with pre-operative or new-onset post-operative AF. Given the paucity of evidence-based information available, clinically relevant TAA-specific research questions have been raised to guide future TAA AF-related investigations.
Keywords
INTRODUCTION
Atrial fibrillation (AF) is among the most frequent arrhythmias, with an estimated global prevalence of more than 33 million cases; within the United States, the estimated AF prevalence ranges from 2.7 to 6.1 million cases[1]. For the North American population, increased AF incidence has been associated with advancing age, male sex, and Caucasian race. Other known AF cardiac-related risk factors include the presence of coronary artery disease, hypertensive heart disease, and valvular disease. Additionally, non-cardiac-related diseases have been associated with increased AF, including diabetes, obesity, and chronic kidney disease[2]. For patients with a history of AF, there is a heightened risk for adverse post-cardiac clinical outcomes, including recurrent AF, embolization, myocardial infarction, stroke, and mortality.
As one of the most common post-cardiac surgery complications, AF incidence has been reported ranging from 10% to 40%; moreover, it is associated with a variety of other post-operative adverse outcomes[3]. Specifically, post-operative AF (POAF) following cardiac surgery has been associated with increased mortality, morbidity, readmission, and overall length of stay.
Recently, there has been a rise in thoracic aortic aneurysms (TAA) incidence reportedly; correspondingly, there has been an increased interest in the use of aortic repair procedures for TAA rupture/dissection patients[4-6]. Given the increased TAA prevalence and treatment rates, it appears timely to review the documented associations between AF and TAA patients’ outcomes.
METHODS
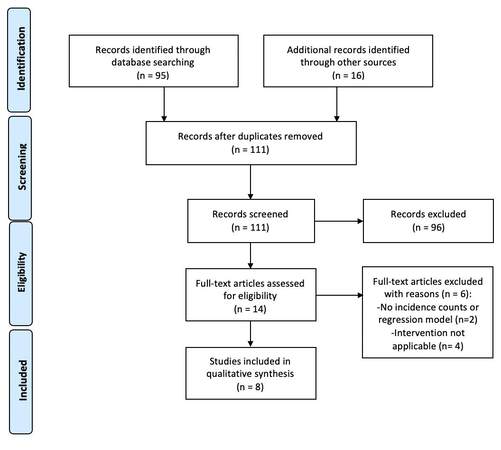
As of September 6, 2021, a comprehensive PubMed literature review was performed. For this Medline search, the extremely broad-based MeSH terms used were “thoracic aortic aneurysm” and “atrial fibrillation”. All initial search articles identified had their abstracts critically appraised by two co-authors (Sohaib Agha and Ashutosh Yaligar) to determine direct relevance to this TAA procedural pre-/post-operative AF topic. Findings from all relevant articles were extracted and summarized. All included articles’ references were also carefully reviewed. Additionally, a Web of Science backward PMID citation search was performed for each included article; all citations were reviewed for potential inclusion based on relevance criteria also. Since only eight directly applicable articles were identified for qualitative analysis, this exhaustive Medline review was intended to present the latest scientific findings available on this topic [Figure 1].
THORACIC AORTIC ANEURYSM RUPTURE/DISSECTION AND SURGICAL INTERVENTIONS
TAAs are usually asymptomatic until a certain threshold size is reached; at a certain point, the risk of TAA dissection or rupture increases. Usually presenting emergently, the complications of TAA rupture and/or dissection carry a high morbidity and mortality rate[7]. According to current TAA literature regarding TAA presentation and/or TAA complications of aortic dissections, the surgical intervention and/or management utilized will be dependent upon the anatomical location of the TAA and whether or not there is a concurrent rupture/dissection. In most cases, surgical intervention involves resection and replacement of the dissected or torn aortic segment; very commonly, these ruptures and dissections present as a surgical emergency.
The DISSECT system has been established to guide clinicians to optimize the management of aortic dissections based upon location, symptom onset, duration of time, and other associated risk factors[8]. Based on DISSECT six characteristics (i.e., disease duration, location of intimal tear, dissected aorta size, aortic involvement extent, dissection-based clinical complications, and aortic false lumen-based thrombi), the clinician is provided contemporary therapeutic approaches to consider. Although not data-driven, this clinical consensus is based on best practices and current knowledge. Interestingly, other patient characteristics (i.e., AF) are not considered as part of the DISSECT system’s risk assessment evaluation. Moreover, the recent TAA guidelines are silent on the topics of pre-operative and post-operative atrial fibrillation[9,10].
PRE-OPERATIVE ATRIAL FIBRILLATION PREVALENCE IN TAA REPAIR
The literature documenting the prevalence of baseline atrial fibrillation in TAA repair patients is scant. Matsuura et al.[9] found that 5% of patients receiving total arch repair for atherosclerotic aortic arch aneurysm or aortic dissection had baseline atrial fibrillation. Another study by Sorabella et al.[10] found that 14.2% of non-octogenarians had baseline AF compared to 26.5% of octogenarians at the time of undergoing aortic root replacement surgery. More research is needed to study the TAA pre-operative AF prevalence and study the impact of pre-operative TAA patients’ AF upon their post-operative outcomes.
POST-OPERATIVE ATRIAL FIBRILLATION [POAF] INCIDENCE IN TAA REPAIR
Specifically focusing on TAA repair, new-onset POAF is significantly associated with increased short-term and long-term adverse outcomes. New-onset POAF in thoracic aortic procedures ranged in incidence from 17.1% to 53.9%[11,12]. Further, multiple studies noted that a large proportion of the new-onset AF population reverts to sinus rhythm (SR) upon discharge or long-term follow-up based upon cardioversion or antiarrhythmic drug therapy. In one study 65.8% (n = 88/134) of new-onset POAF patients returned to SR[13] as compared to another study that noted 100% (n = 33) TAA patients with new-onset POAF returned to sinus rhythm[13]. Incidence rates of POAF from population-based studies are summarized in [Table 1].
Incidence rates of POAF literature summary of retrospective studies
| Author | Sample size (n) | Surgery type/location | Post-operative AF (%) |
| Arakawa et al.[14] | 12,260 | Root (1347) Ascending (5588) Arch (5161) Descending (2332) Thoracoabdominal (932) | Root (20.4%) Ascending (21.2%) Arch (20.9%) Descending (10.5%) Thoracoabdominal (8.5%) |
| Compostella et al.[15] | Group A: 67 | Graft replacement of ascending aorta (67) | Graft replacement of ascending aorta: 29 pt (43%) |
| He et al.[11] | 126 | Total: 126 Ascending aorta (88) Aortic arch (61) Thoracoabdominal (29) | Total: 26.2% Ascending aorta (31.8%) Aortic arch (32.8%) Thoracoabdominal (13.8%) |
| Matsuura et al.[9] | 459 | Total aortic arch repair (459) | Total aortic arch repair (52.7%) |
| Matsuura et al.[12] | 445 | Aortic arch surgery (445) | Aortic arch surgery (53.9%) |
|
Perzanowski et al.[16] | 153 | Thoracic aorta (63) Abdominal aorta (90) | Total: 30 pt Thoracic aorta: 21 pt (33%) Abdominal aorta: 9 pt (10%) |
TAA PATIENTS’ RISK FACTORS PREDISPOSING TO POAF
As many as fifty percent of patients undergoing thoracic surgery develop new-onset POAF[11]. Given the high prevalence of this adverse outcome, researchers have studied whether any baseline characteristics are associated with its development. Matsuura et al.[9] studied patients undergoing total arch repair for atherosclerotic aortic arch aneurysm or aortic dissection, and found that advanced age was the primary risk factor that correlated with post-operative atrial fibrillation development. He et al.[11] studied patients with aortic aneurysm, or dissection of the ascending, arch, thoracic, or abdominal aorta; their study again documented advanced age as an important risk characteristic; in multivariable analysis, however, cardiac dysfunction was an independent POAF predictor in addition to older age.
Nishi et al.[13] evaluated Japanese patients in their Adult Cardiac Surgery database; of these patients receiving a variety of cardiac surgical procedures, patient age [odds ratio (OR) = 1.55 for each decade with 95% confidence interval (CI) from 1.30 to 1.85], any blood product transfusion (OR = 2.09, 95%CI: 1.03 to 4.26), and a thoracic aortic procedure (OR = 2.08, 95%CI: 2.12 to 3.55) were the most important multivariable model POAF predictors.
Other studies have examined the impact of TAA patient’s baseline risk factors on the development of new-onset POAF, including age, history of smoking, operation urgency, chronic obstructive pulmonary disease, diabetes, hypertension, perioperative myocardial infarction, thoracic surgery, and concomitant non-elective coronary artery bypass graft (CABG) surgery[12,14-16]. Compared to abdominal aortic procedures, Perzanowski et al.[16] identified, using multivariable modeling, thoracic aortic repair procedures as particularly vulnerable to POAF, as well as patients with thoracic aortic repairs who were also hypertensive represented a special “at risk” patient sub-group. See [Table 2] for the list of multivariable risk factors that were statistically significant POAF predictors.
Literature summary of retrospective studies
| Author | Univariate predictors of POAF | Multivariate predictors of POAF |
| Arakawa et al.[14] | - Age - History of smoking - Congestive heart failure - Urgent operation - Concomitant with non-elective CABG | (None identified) |
| Compostella et al.[15] | - Older age | (None identified) |
| He et al.[11] | - Advanced age - Female sex - Cardiac dysfunction - Operation on ascending aorta | - Advanced age - Cardiac dysfunction |
| Matsuura et al.[9] | -Advanced age | (None identified) |
| Matsuura et al.[12] | -Increased input | (None identified) |
| Perzanowski et al.[16] | - Age - Diabetes - Hypertension - Chronic pulmonary disease - Perioperative myocardial infarction - Thoracic surgery | - Age - Diabetes - Thoracic surgery |
OUTCOMES OF POST-OPERATIVE ATRIAL FIBRILLATION (POAF) IN TAA REPAIR
The outcomes of post-operative atrial fibrillation in TAA repair are favorable, depending upon how early TAA management measures are implemented. One of the best-known prophylactic treatments for atrial fibrillation is B-blocker therapy, and this same treatment has also been utilized for post-operative atrial fibrillation[13]. Interestingly, however, no significant difference in POAF rates was documented between TAA patients with and without pre-operative beta-blocker therapy. A study was conducted that compared outcomes following aortic root replacement in octogenarians versus non-octogenarians. It was found that octogenarians could safely undergo this surgery with moderately worse but acceptable perioperative mortality and late survival. Several post-operative complications were reported, including POAF and reoperation for bleeding, stroke, respiratory failure, new need for continuous renal replacement therapy, myocardial infarction, and new permanent pacemaker. The octogenarians had a significantly higher rate of POAF (60.6% octogenarians versus 38.5% non-octogenarians, P < 0.01). Even though there was a higher octogenarian POAF rate, there was no statistically significant difference in the post-TAA procedural mortality or post-operative complication rates[11].
In a Japanese bivariate analysis of 12,660 TAA procedures across 244 hospitals, new-onset POAF was found by Arakawa et al.[14] to be associated with increased risk of 30-day operative mortality (i.e., defined as death within 30 days or in-hospital death); the POAF 30-day operative mortality rate was 10.1% versus the non-POAF patients’ rate of 6.6% (P < 0.01). There were dramatic differences in the POAF rates by TAA-related procedures ranging from ascending aortic surgery with the highest POAF rate (21.2%; n = 1185/5588) to thoracoabdominal aortic surgery (8.5%; n = 79/932). Across all TAA procedures, the overall POAF rate was 17.1% (n = 2095/12,260). Additionally, this TAA study identified the statistically significant predictors (p < 0.05) for POAF; these predictors included acute aortic events (i.e., dissection or rupture), advanced age, body surface area, urgent surgical status, concomitant CABG procedure, concomitant valve procedure, aortic root procedures ascending aortic arch procedures, and arch procedures. Additionally, preoperative antiarrhythmic drugs (i.e., digitalis, beta-blockers, or nitrates) did not seem to impact POAF rates[14]. In Perzanowski’s study, the patients that developed new-onset POAF had higher rates of post-operative myocardial infarctions (25%) vs. non-POAF patients (10%). Further, new-onset POAF has been associated in studies with increased length of post-operative hospital stay and increased length of ICU stay. In their Japanese single-center study from 1993 to 2004, Matsuura et al.[9] identified length of stay for POAF patients as 49 ± 76 days versus non-POAF patients of 12 ± 23 days, P < 0.01. Most importantly, this publication concluded that good POAF patient management was required in order to assure good TAA patients’ outcomes[15].
INSIGHTS FROM PRACTICING CLINICIANS
Overall, it is quite remarkable how few publications have addressed the perennial AF problem for the fast-growing TAA patient population. Remarkably, no systematic literature reviews were identified on this TAA POAF topic. In the two recently published guidelines by Society of Thoracic Surgeons/American Association Thoracic Surgeons (STS/AATS) regarding Type A and B dissections, pre-operative and post-operative AF topics were not addressed[17-19]. As a concern, cardiac tamponade, acute or delayed, arising from prolonged bleeding represents a constant challenge. Hence, there is a desire to avoid or delay anticoagulation therapy until it is no longer possible. Given the lack of information on how to define, prevent, and treat TAA patients with AF challenges, several clinical care questions remain outstanding:
● Should all TAA patients receive prophylactic medications to prevent new-onset POAF?
● For the sub-group of TAA patients with pre-op AF or POAF:
■ How long should anticoagulation be delayed?
■ What anticoagulation agents should be used as first-line agents?
■ What mode of administration is generally preferable (oral or intravenous)?
● For the subgroup of TAA patients with POAF:
■ Is immediate cardioversion (i.e., rhythm control) advantageous to consider?
Although this approach has been commonly used, there is randomized trial evidence for a general cardiac surgical population documenting that there is no advantage to rhythm control over rate control[20]. In the most recent 2021 clinical practice guidelines reported for surgical treatment of acute type A and B aortic dissection[17-19], no mention is made of either pre-operative TAA AF-related risks or post-TAA POAF as a major complication requiring attention; why is this discussion missing? Given other recent guideline recommendations for first-line vs. second-line medical therapies, additional recommendations for prophylaxis in this particular high-risk TAA patient population would be welcomed.
Importantly, the absence of these latest clinical practice guidelines addressing the topic of arrhythmia complications appears to be very telling: POAF as a TAA complication has not yet hit the cardiothoracic professional society “quality improvement program” radar screen. Coming from an era where short-term survival was the foremost concern, POAF is still considered a minor annoyance–rather than a potentially life-altering event with serious implications for future adverse clinical outcomes and increased costs. Now that these patients survive longer, entirely overlooking the topic of arrhythmias, as the most frequently occurring post-TAA complication, seems no longer tenable and justifies the development of pre-TAA/post-TAA POAF-focused consensus standards.
CONCLUSION
For TAA procedures, the most common place complication occurring is POAF. For the published literature as of September 6, 2021, this comprehensive literature review identified only a handful of articles reporting pre-/post-TAA AF rates. Of these, the TAA-specific risk factors associated with increased POAF included: older age, gender, cardiac dysfunction, and the type of aortic procedure (e.g., aortic arch repairs). In this search, several historical risk factors associated with POAF for other cardiac surgical procedures had not yet been associated with the post-TAA procedural risk of POAF. The missing POAF-related traditional risk factors included hypertension, coronary artery disease, diabetes, and congestive heart failure. To date, moreover, no thoracic endovascular aortic repair publications were found on this pre-operative AF or POAF topic; thus, this lack of evidence-based literature to drive future TAA patient care represents a very serious clinical concern.
Given the paucity of TAA AF publications, pre- and post-TAA AF rates were not documented in the contemporaneous literature; moreover, TAA patients’ rates for post-discharge AF recurrences were not reported. Given the lack of TAA patients’ data-driven AF findings, clinicians have very little evidence to establish their expectations for internal TAA quality assurance initiatives. Importantly, TAA POAF prophylactic and treatment strategies have not yet been thoroughly evaluated. No TAA-focused patient clinical trials were found to document the effectiveness of standard POAF prophylactic approaches or POAF-related treatments (e.g., beta-blocker use pre-operatively)[13] used in other cardiac surgical patient populations. Similarly, comparisons of POAF rate control vs. rhythm control strategies have not yet been published focused specifically on TAA patients. Due to the lack of TAA-specific AF studies, clinicians treating TAA patients will need to proceed in the interim applying the research findings published for other cardiac surgical patient populations.
TAA repair or arch replacement procedures are at high risk for bleeding complications; therefore, efforts to avoid post-TAA POAF anticoagulation are now opportune for future investigation. In other cardiac surgery populations (e.g., CABG patients), specific POAF treatments (i.e., beta-blockers, amiodarone) have been documented to significantly reduce POAF rates[18]. Although beta-blocker appropriateness has not yet been rigorously evaluated for TAA-related procedures, the recent clinical practice standards include this strategy. In light of the promotion of recent TAA clinical practice guidelines, this lack of data-driven evidence for specific POAF therapies for TAA patients should raise a call to arms.
In conclusion, a TAA knowledge chasm exists regarding atrial fibrillation patients’ care. Given this challenge, additional TAA patient research appears urgently needed to identify, manage, and improve the future quality of care for this vulnerable, post-TAA POAF “at risk” patient population.
DECLARATIONS
Author’s contributionsStudy conception and design: Kuruvilla AS, Agha S, Yaligar A, Tannous HJ, McLarty AJ, Shroyer AL, Bilfinger TV
Acquisition of data: Kuruvilla AS, Agha S, Yaligar A, Tannous HJ, McLarty AJ, Shroyer AL, Bilfinger TV
Analysis and interpretation of data: Kuruvilla AS, Agha S, Yaligar A, Tannous HJ, McLarty AJ, Shroyer AL, Bilfinger TV
Drafting of manuscript: Kuruvilla AS, Agha S, Yaligar A, Tannous HJ, McLarty AJ, Shroyer AL, Bilfinger TV
Critical revision: Kuruvilla AS, Agha S, Yaligar A, Tannous HJ, McLarty AJ, Shroyer AL, Bilfinger TV
Final approval: Kuruvilla AS, Agha S, Yaligar A, Tannous HJ, McLarty AJ, Shroyer AL, Bilfinger TV
Availability of data and materialsData source: PubMed and Web of Science
Financial support and sponsorshipNone.
Conflicts of interestAll authors declared that there are no conflicts of interest.
Ethical approval and consent to participateNot applicable.
Consent for publicationNot applicable.
Copyright© The Author(s) 2022.
REFERENCES
1. Chung MK, Eckhardt LL, Chen LY, et al. American Heart Association Electrocardiography and Arrhythmias Committee and Exercise. Lifestyle and risk factor modification for reduction of atrial fibrillation: a scientific statement from the American heart association. Circulation 2020;141:e750-72.
2. Anumonwo JM, Kalifa J. Risk factors and genetics of atrial fibrillation. Cardiol Clin 2014;32:485-94.
3. Almassi GH, Schowalter T, Nicolosi AC, et al. Atrial fibrillation after cardiac surgery: a major morbid event? Ann Surg 1997;226:501-11;discussion 511.
4. McClure RS, Brogly SB, Lajkosz K, Payne D, Hall SF, Johnson AP. Epidemiology and management of thoracic aortic dissections and thoracic aortic aneurysms in Ontario, Canada: a population-based study. J Thorac Cardiovasc Surg 2018;155:2254-2264.e4.
5. Olsson C, Thelin S, Ståhle E, Ekbom A, Granath F. Thoracic aortic aneurysm and dissection: increasing prevalence and improved outcomes reported in a nationwide population-based study of more than 14,000 cases from 1987 to 2002. Circulation 2006;114:2611-8.
6. Wang GJ, Jackson BM, Foley PJ, et al. National trends in admissions, repair, and mortality for thoracic aortic aneurysm and type B dissection in the national inpatient sample. J Vasc Surg 2018;67:1649-58.
7. Goldfinger JZ, Halperin JL, Marin ML, Stewart AS, Eagle KA, Fuster V. Thoracic aortic aneurysm and dissection. J Am Coll Cardiol 2014;64:1725-39.
8. Alfson DB, Ham SW. Type B aortic dissections: current guidelines for treatment. Cardiol Clin 2017;35:387-410.
9. Matsuura K, Ogino H, Matsuda H, et al. Prediction and incidence of atrial fibrillation after aortic arch repair. Ann Thorac Surg 2006;81:514-8.
10. Sorabella RA, Wu YS, Bader A, et al. Aortic root replacement in octogenarians offers acceptable perioperative and late outcomes. Ann Thorac Surg 2016;101:967-72.
11. He Y, Peng H, Jian K, Li Q, Liu W, Li Y. Incidence of atrial fibrillation and risk factors analyzing in 126 cases of aortic surgery. Cardiology Plus 2018;3:127-31.
12. Matsuura K, Ogino H, Matsuda H, et al. Impact of volume status on the incidence of atrial fibrillation following aortic arch repair. Heart Vessels 2007;22:21-4.
13. Nishi H, Sakaguchi T, Miyagawa S, et al. Atrial fibrillation occurring early after cardiovascular surgery: impact of the surgical procedure. Surg Today 2013;43:1134-9.
14. Arakawa M, Miyata H, Uchida N, et al. Postoperative atrial fibrillation after thoracic aortic surgery. Ann Thorac Surg 2015;99:103-8.
15. Compostella L, Russo N, D'Onofrio A, et al. Abnormal heart rate variability and atrial fibrillation after aortic surgery. Rev Bras Cir Cardiovasc 2015;30:55-62.
16. Perzanowski C, Gandhi S, Pai RG. Incidence and predictors of atrial fibrillation after aortic repairs. Am J Cardiol 2004;93:928-30.
17. Malaisrie SC, Szeto WY, Halas M, et al. AATS Clinical Practice Standards Committee: Adult Cardiac Surgery. 2021 the American association for thoracic surgery expert consensus document: surgical treatment of acute type A aortic dissection. J Thorac Cardiovasc Surg 2021;162:735-758.e2.
18. Crystal E, Connolly SJ, Sleik K, Ginger TJ, Yusuf S. Interventions on prevention of postoperative atrial fibrillation in patients undergoing heart surgery: a meta-analysis. Circulation 2002;106:75-80.
19. Michelena HI, Della Corte A, Evangelista A, et al. Endorsed by the Heart Valve Society (HVS). International consensus statement on nomenclature and classification of the congenital bicuspid aortic valve and its aortopathy, for clinical, surgical, interventional and research purposes. Eur J Cardiothorac Surg 2021;60:448-76.
Cite This Article
Export citation file: BibTeX | RIS
OAE Style
Kuruvilla AS, Agha S, Yaligar A, Tannous HJ, McLarty AJ, Shroyer AL, Bilfinger TV. A literature review: pre-/post-operative atrial fibrillation for thoracic aortic aneurysm procedures. Vessel Plus 2022;6:45. http://dx.doi.org/10.20517/2574-1209.2021.122
AMA Style
Kuruvilla AS, Agha S, Yaligar A, Tannous HJ, McLarty AJ, Shroyer AL, Bilfinger TV. A literature review: pre-/post-operative atrial fibrillation for thoracic aortic aneurysm procedures. Vessel Plus. 2022; 6: 45. http://dx.doi.org/10.20517/2574-1209.2021.122
Chicago/Turabian Style
Kuruvilla, Annet S., Sohaib Agha, Ashutosh Yaligar, Henry J. Tannous, Allison J. McLarty, A. Laurie Shroyer, Thomas V. Bilfinger. 2022. "A literature review: pre-/post-operative atrial fibrillation for thoracic aortic aneurysm procedures" Vessel Plus. 6: 45. http://dx.doi.org/10.20517/2574-1209.2021.122
ACS Style
Kuruvilla, AS.; Agha S.; Yaligar A.; Tannous HJ.; McLarty AJ.; Shroyer AL.; Bilfinger TV. A literature review: pre-/post-operative atrial fibrillation for thoracic aortic aneurysm procedures. Vessel Plus. 2022, 6, 45. http://dx.doi.org/10.20517/2574-1209.2021.122
About This Article
Special Issue
Copyright
Data & Comments
Data

 Cite This Article 9 clicks
Cite This Article 9 clicks










Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at support@oaepublish.com.