Transthoracic vs. transesophageal echocardiography in transcatheter aortic valve implantation: a systematic review and meta-analysis
Abstract
Aim: Traditionally, transcatheter aortic valve implantation (TAVI) was performed under general anesthesia (GA) accompanied by intraprocedural transesophageal echocardiography (TEE). Recently, minimalist TAVI with monitored anesthesia care (MAC) and transthoracic echocardiography (TTE) has gained popularity. However, TTE imaging quality may be suboptimal compared to TEE and may increase the risks of paravalvular leak (PVL). We sought to compare TTE to TEE for PVL (primary outcome) and secondary safety outcomes in a study-level meta-analysis.
Methods: Ovid versions of Medline and Embase were searched from 1946 to 2018 for studies comparing TTE to TEE in TAVI directly or MAC to GA in TAVI (must also specify echocardiography usage) and meta-analyzed in a random effects model.
Results: Sixteen studies (n = 3,510) were included in the meta-analysis. The rate of any PVL was not significantly different between TTE-TAVI and TEE-TAVI groups (18.4% vs. 21.4%, risk ratio: 1.01, 95%CI: 0.83 to 1.23, P = 0.92, I2 = 36%). Similarly, there were no significant differences in secondary safety outcomes. Resource utilization was lower with TTE-TAVI; hospital LOS [mean difference (MD): -1.55 days, 95%CI: -2.27 to -0.83, P < 0.01], contrast volume (MD: -24.75 mL, 95%CI: -49.48 to -0.03, P = 0.05) and procedure time (MD: -31.09 min, 95%CI: -54.77 to -7.40, P < 0.01) were significantly lower.
Conclusion: The use of TTE-TAVI in conjunction with MAC was not associated with an increased risk of PVL and was associated with lower hospital resource utilization. However, other factors, such as mode of anesthesia, may have influenced these findings.
Keywords
Introduction
The volume of transcatheter aortic valve implantation (TAVI) has surpassed surgical aortic valve replacement in Europe and North America[1-3]. Traditionally, TAVI uses general anesthesia (GA) accompanied by intraprocedural transesophageal echocardiography (TEE) to assess root geometry and guide valve placement during the procedure[4]. While rare, TEE can also lead to serious complications such as esophageal perforation, aspiration and oropharyngeal damage[5].
Recently, minimalist TAVI conducted under monitored anesthesia care (MAC) has gained popularity. Studies suggest that it is less invasive and allows for earlier mobilization and patient discharge without compromising early outcomes[6]. Since TEE is not usually conducted under MAC, minimalist TAVI often uses transthoracic echocardiography (TTE) instead[5]. Image quality with TTE may be suboptimal compared to TEE and may have a lower sensitivity in detecting paravalvular leaks (PVL)[5,7-8]. Studies have shown that PVL after TAVI is often poorly tolerated and associated with excess late mortality[8,9]. Thus, understanding whether PVL increases in patients undergoing TAVI with TTE compared to TEE is paramount. There are few studies directly comparing TTE and TEE in TAVI and the sample size in these studies are limited. However, there are many studies that compare usage of MAC and GA in TAVI[6]. Since TTE and TEE usage is closely associated with the mode of anesthesia, MAC and GA can be used as surrogate variables for TTE and TEE respectively.
This quantitative meta-analysis aims to compare early echocardiography parameters and safety outcomes in TTE-TAVI and TEE-TAVI. The primary outcome is PVL and secondary outcomes include 30-day mortality, renal failure, stroke, major bleeds, hospital length of stay (LOS), ICU LOS, fluoroscopy time, contrast volume and procedure time.
Methods
Systematic literature review
Ovid versions of Medline and Embase were searched from 1946 to June 18, 2018 for the following key terms: “Transcatheter aortic valve implantation,” “echocardiography” and “anesthesia”. The full search strategy is shown in Supplementary Appendix S1. Inclusion criteria included the following: English, comparison of TTE to TEE or MAC to GA in TAVI, reported at least 1 chosen outcome. MAC vs GA papers must also specify the frequency of TTE and TEE usage. Exclusion criteria included the following: non-English studies, conference abstracts, proceedings, case reports and other non-comparative study designs. The citations from the literature search were compared to papers in a meta-analysis comparing MAC to GA in TAVI to ensure papers were not missed[6].
All titles and abstracts were reviewed. Full papers of citations that could potentially be included in the study were further analyzed. The literature search results were reviewed independently by 2 investigators (T.L. and A.D.). Disagreements were discussed by investigators until an agreement was reached.
Quality assessment and data abstraction
Quality of studies were assessed with the Grading of Recommendations Assessment, Development and Evaluation approach[10]. The following outcomes were abstracted: usage of TTE, usage of TEE, mild PVL, moderate PVL, severe PVL, any PVL, 30-day mortality, renal failure, stroke, major bleeds, hospital LOS, ICU LOS, fluoroscopic time, contrast volume, and procedure time.
Analysis
A random effects meta-analysis was performed with R packages meta and metafor (R version 3.5.0; R Foundation for Statistical Computing, Vienna, Austria). If only the median and quartiles were provided in a study, mean and standard deviation were estimated based on protocol found in the literature[11,12]. Risk ratios (RRs) weighted based on number of events and sample size were calculated using the Mantel-Haenszel method for perioperative dichotomous outcomes. The event rates and percentages for all subgroups were calculated based on raw data for perioperative dichotomous outcomes. Mean differences (MD) were used to analyze continuous variables as calculated by the inverse variance method. Means were also calculated based on raw data for continuous variables. Heterogeneity was reported as low (I2 = 0%-25%), moderate (I2 = 26%-50%), or high (I2 > 50%), as reported previously in the literature[13]. Data was analyzed as a pooled group then as 2 sub-groups: matched and unmatched observational studies.
One additional post hoc analysis was conducted. It compared studies that only used TTE in the MAC group to studies that had mixed TTE and TEE usage in the MAC group on the outcome of PVL.
Results
After searching in Ovid and Embase, 1,133 citations were screened. Sixteen citations (n = 3510 patients) fulfilled the inclusion and exclusion criteria. There were 10 unmatched (n = 2037 patients)[7,14-22] and 6 matched (n = 1473 patients)[4,23-27] retrospective observational studies [Supplementary Figure 1].
The studies were rated at low or moderate risk of bias [Supplementary Table 1]. All studies had concurrent controls. Twelve studies[4,14-18,20,21,24-27] did not report follow up rates. One study lost more than 10% of patients in the 1 year follow-up[7]. TTE-TAVI and TEE-TAVI shared similar baseline characteristic, including predicted mortality risk scores [Supplementary Tables 2 and 3].
Primary outcome: PVL
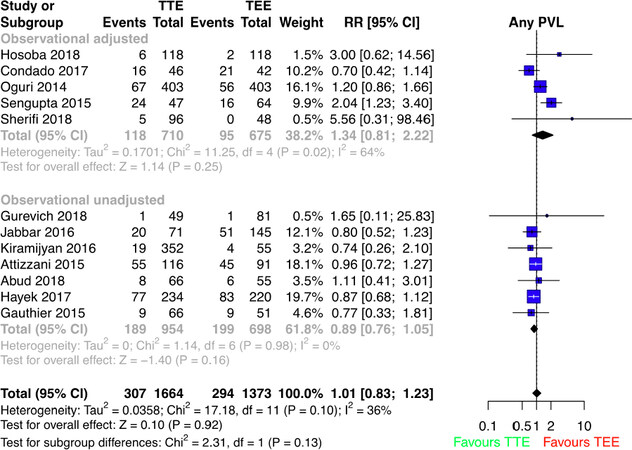
Event rates and relative risks for the primary outcome and periprocedural events are summarized in Table 1. Corresponding forest plots are shown in Figure 1 and Supplementary Figures 2-8. The incidence rate in pooled results for mild PVL was not significantly different when TTE-TAVI was compared to TEE-TAVI (32.1% vs. 34.0%, RR: 0.95, 95%CI: 0.72 to 1.25, P = 0.71, I2 = 54%). Similar non-significant results were found for moderate PVL (6.6% vs. 6.7%, RR: 0.87, 95%CI: 0.55 to 1.38, P = 0.56, I2 = 0%), severe PVL (0.9% vs. 0.3%, RR: 1.39, 95%CI: 0.06 to 33.21, P = 0.84, I2 = 53%), and any PVL ³ mild (18.4% vs. 21.4%, RR: 1.01, 95%CI: 0.83 to 1.23, P = 0.92, I2 = 36%). All subgroups also did not show any significant difference between TTE-TAVI and TEE-TAVI.
Pooled event rates and relative risks for dichotomous events from meta-analysis
| TTE event rate | TEE event rate | RR (95%CI) | P value | |
|---|---|---|---|---|
| Mild PVL | ||||
| Observational adjusted | 33/93 (35.5%) | 33/106 (31.1%) | 1.09 (0.4 to 2.95) | 0.87 |
| Observational unadjusted | 132/421 (31.4%) | 158/456 (34.6%) | 0.89 (0.73 to 1.08) | 0.23 |
| Pooled | 165/514 (32.1%) | 191/562 (34.0%) | 0.95 (0.72 to 1.25) | 0.71 |
| Moderate PVL | ||||
| Observational adjusted | 3/47 (6.4%) | 0/64 (0.0%) | 9.48 (0.5 to 179.24) | 0.13 |
| Observational unadjusted | 32/482 (6.6%) | 32/417 (7.7%) | 0.82 (0.52 to 1.31) | 0.41 |
| Pooled | 35/529 (6.6%) | 32/481 (6.7%) | 0.87 (0.55 to 1.38) | 0.56 |
| Severe PVL | ||||
| Observational adjusted | 3/350 (0.9%) | 1/311 (0.3%) | 1.39 (0.06 to 33.21) | 0.84 |
| Observational unadjusted | N/A | |||
| Pooled | 3/350 (0.9%) | 1/311 (0.3%) | 1.39 (0.06 to 33.21) | 0.84 |
| Any PVL | ||||
| Observational adjusted | 118/710 (16.6%) | 95/675 (14.1%) | 1.34 (0.81 to 2.22) | 0.25 |
| Observational unadjusted | 189/954 (19.8%) | 199/698 (28.5%) | 0.89 (0.76 to 1.05) | 0.16 |
| Pooled | 307/1,664 (18.4%) | 294/1,373 (21.4%) | 1.01 (0.83 to 1.23) | 0.92 |
| 30-day mortality | ||||
| Observational adjusted | 5/90 (5.6%) | 5/86 (5.8%) | 0.96 (0.28 to 3.27) | 0.94 |
| Observational unadjusted | 37/955 (3.9%) | 31/628 (4.9%) | 0.65 (0.37 to 1.11) | 0.11 |
| Pooled | 42/1,045 (4.0%) | 36/714 (5.0%) | 0.69 (0.49 to 1.13) | 0.14 |
| Renal Failure | ||||
| Observational adjusted | 20/707 (2.8%) | 10/655 (1.5%) | 1.42 (0.57 to 3.55) | 0.45 |
| Observational unadjusted | 59/760 (7.8%) | 36/486 (7.4%) | 1.61 (0.34 to 7.7) | 0.18 |
| Pooled | 79/1,467 (5.4%) | 46/1,141 (4.0%) | 0.86 (0.58 to 1.26) | 0.43 |
| Stroke | ||||
| Observational adjusted | 15/614 (2.4%) | 15/627 (2.4%) | 1.01 (0.49 to 2.07) | 0.99 |
| Observational unadjusted | 26/834 (3.1%) | 17/555 (3.1%) | 0.73 (0.37 to 1.44) | 0.36 |
| Pooled | 41/1,448 (2.8%) | 32/1,182 (2.7%) | 0.85 (0.52 to 1.39) | 0.52 |
| Major bleed | ||||
| Observational adjusted | 23/567 (4.1%) | 39/563 (6.9%) | 0.48 (0.13 to 1.7) | 0.25 |
| Observational unadjusted | 12/689 (1.7%) | 9/382 (2.4%) | 0.79 (0.31 to 2.05) | 0.63 |
| Pooled | 35/1,256 (2.8%) | 48/945 (5.1%) | 0.58 (0.28 to 1.22) | 0.15 |
Figure 1. Forest plot for relative risk ratios of any PVL for TTE-TAVI in comparison to TEE-TAVI with subgroups based on study type. TTE; transthoracic echocardiography; TEE: transesophageal echocardiography; RR: relative risk; CI: confidence interval; PVL: paravalvular leak; TAVI: transcatheter aortic valve implantation
Periprocedural events
When comparing patients that had TTE and TEE at the time of TAVI, there was no significant difference in pooled event rates for 30-day mortality (4.0% vs. 5.0%, RR: 0.69, 95%CI: 0.49 to 1.13, P = 0.14, I2 = 0%) [Table 1]. Similarly, when TTE-TAVI was compared to TEE-TAVI, no significant differences were found for renal failure (5.4% vs. 4.0%, RR: 0.86, 95%CI: 0.58 to 1.26, P = 0.43, I2 = 0%), stroke (2.8% vs. 2.7%, RR: 0.85, 95%CI: 0.52 to 1.39, P = 0.52, I2 = 0%), and major bleed (2.8% vs. 5.1%, RR: 0.58, 95%CI: 0.28 to 1.22, P = 0.15, I2 = 45%) [Table 1]. All subgroups also did not show a significant difference when TTE-TAVI was compared to TEE-TAVI.
Hospital resources
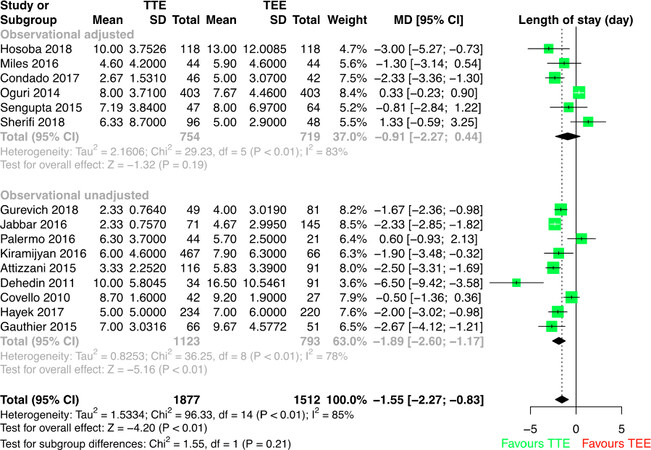
The weighted means and MDs for the continuous outcomes are summarized in Table 2. Corresponding forest plots are depicted in Figure 2 and Supplementary Figures 9-12. Hospital LOS was significantly shorter in the TTE-TAVI group compared to the TEE-TAVI group (MD: -1.55 days, 95%CI: -2.27 to -0.83, P ≤ 0.01). However, hospital LOS was significantly lower in unmatched observational studies (MD: -1.89 days, 95%CI: -2.6 to -1.17, P ≤ 0.01) but equivalent in matched observational studies (MD: -0.91 days, 95%CI: -2.27 to 0.44, P = 0.19).
Pooled event means and weighted differences for continuous events from meta-analysis
| TTE mean | TEE mean | MD (95%CI) | P value | |
|---|---|---|---|---|
| Length of stay (days) | ||||
| Observational adjusted | 6.3 | 7.2 | -0.91 (-2.27 to 0.44) | 0.19 |
| Observational unadjusted | 5.3 | 7.2 | -1.89 (-2.6 to -1.17) | < 0.01 |
| Pooled | 5.6 | 7.2 | -1.55 (-2.27 to -0.83) | < 0.01 |
| ICU time (h) | ||||
| Observational adjusted | 59.7 | 54.2 | 6.22 (-16.34 to 28.78) | 0.59 |
| Observational unadjusted | 39.6 | 59.5 | -12.68 (-22.28 to -3.09) | < 0.01 |
| Pooled | 47.4 | 49.9 | -5.6 (-13.66 to 2.45) | 0.17 |
| Fluoroscopic time (min) | ||||
| Observational adjusted | 20.9 | 21.7 | -0.71 (3.91 to 2.5) | 0.67 |
| Observational unadjusted | 21.0 | 22.8 | -1.79 (-3.36 to -0.22) | 0.03 |
| Pooled | 21.0 | 22.3 | -1.34 (-2.84 to 0.16) | 0.08 |
| Contrast volume (mL) | ||||
| Observational adjusted | 126.7 | 161.7 | -35.0 (-56.89 to -13.11) | < 0.01 |
| Observational unadjusted | 96.2 | 119.0 | -22.83 (-51.14 to 5.48) | 0.11 |
| Pooled | 101.2 | 126.0 | -24.75 (-49.48 to -0.03) | 0.05 |
| Procedure time (min) | ||||
| Observational adjusted | 98.6 | 118.1 | -44.29 (-90.96 to 2.38) | < 0.06 |
| Observational unadjusted | 123.1 | 142.6 | -19.44 (-33.01 to -5.87) | < 0.01 |
| Pooled | 110.5 | 130 | -31.09 (-54.77 to -7.40) | 0.01 |
Figure 2. Forest plot for weighted mean differences (days) of length of stay for TTE-TAVI in comparison to TEE-TAVI with subgroups based on study type. TTE: transthoracic echocardiography; TEE: transesophageal echocardiography; MD: mean difference; CI: confidence interval; TAVI: transcatheter aortic valve implantation
ICU LOS was not significantly different in the TTE group in comparison to the TEE group (MD: -5.6 h, 95%CI: -13.66 to 2.45, P = 0.17). ICU LOS was significantly lower in unmatched observational studies (MD: -12.68 h, 95%CI: -22.28 to -3.09, P ≤ 0.01) but equivalent in matched observational studies (MD: 6.22 h, 95%CI: -16.34 to 28.78, P = 0.59).
Fluoroscopic time was not significantly different in pooled results when TTE-TAVI was compared to TEE-TAVI (MD: -1.34 min, 95%CI: -2.84 to 0.16, P = 0.08). Likewise, fluoroscopic time was significantly reduced in unmatched observational studies (MD: -1.79 min, 95%CI: -3.36 to -0.22, P = 0.03) but equivalent in matched observational studies (MD: -0.71 min, 95%CI: 3.91 to 2.5, P = 0.67).
Analysis of the data showed that contrast volume was significantly higher in the TTE group (MD: -24.75 mL, 95%CI: -49.48 to -0.03, P = 0.05). However, this trend was driven by the matched observational subgroup, which was significant (MD: -35.00 mL, 95%CI: -56.89 to -13.11, P ≤ 0.01), and not significant in the unmatched observational subgroup (MD: -22.83 mL, 95%CI: -51.14 to 5.48, P = 0.11).
The procedure time was significantly shorter in the TTE group compared to the TEE group (MD: -31.09 min, 95%CI: -54.77 to -7.40, P ≤ 0.01). The subgroups also showed similar patterns.
Post hoc analyses
Any PVL rates between TTE-TAVI and TEE-TAVI were compared in a subgroup analysis of MAC groups that only used TTE vs. MAC groups that had mixed TTE and TEE usage. There were 909 patients in the 100% TTE group and 755 patients in the mixed group. In the mixed echocardiography usage group, the mean use of TTE in the TAVI with MAC group was 50.0%. There were similar crude rates of PVL between the 100% TTE group compared to the mixed echocardiography usage group (18.4% vs. 21.4%). There were no significant differences of PVL between the 100% TTE group (RR: 1.00, 95%CI: 0.79 to 1.26, P = 0.98) and mixed echocardiography group (RR: 1.15, 95%CI: 0.84 to 1.57, P = 0.39, interaction P = 0.49) [Supplementary Figure 13].
Discussion
This is the first meta-analysis using method of anesthesia to infer TTE and TEE status in conjunction with papers that directly compare TTE-TAVI with MAC and TEE-TAVI with GA. The rate of any PVL was not significantly different between TTE-TAVI with MAC and TEE-TAVI with GA. There was also no significant difference found for mild, moderate and severe PVL between the two groups. Importantly, there were no significant differences in safety outcomes such as 30-day mortality and complication rates. Resource utilization was lower with TTE use in conjunction with MAC; hospital LOS, contrast volume and procedure time were reduced. These results are important because the acute nature of post-TAVI PVL is poorly tolerated by patients; even mild PVL leads to higher early and late mortality[9,28]. Existing post-TAVI PVL treatments have not been widely validated[9]. For this reason, prophylactic measures to prevent PVL is paramount to safe TAVI procedures.
Of the 16 papers included in this meta-analysis, only 2 directly compared TTE and TEE[7,27]. Our meta-analysis narrowed the gap in the literature by using a surrogate variable, which enabled us to use an additional 5 matched and 9 unmatched observational studies for a total of 3150 patients. Similar to the previous 2 studies investigating echocardiography in TAVI, the Hayek et al.[7] and Sherifi et al.[27] (propensity matched) papers did not find significant differences in PVL complications between TTE-TAVI and TEE-TAVI groups[7,27]. However, this meta-analysis provides additional insight because it combines all available studies on this topic, allowing us to increase the sample size and power to examine for any differences.
Since TTE-TAVI with MAC was associated with shorter hospital LOS and procedure time, TTE-TAVI with MAC appeared to utilize less hospital resources without any increase in peri-operative morbidity and mortality. However, other factors may affect cost of procedure. Hayek et al.[7] found that TTE-TAVI was associated with significantly higher rates of second valve implantations (odds ratio = 1.58, 95%CI: 1.01 to 2.46), which may negate the beneficial effects of shorter LOS and procedure time on cost. Hence, a more detailed cost analysis will need to be conducted to determine the economic benefits of anesthesia and echocardiography type at the time of TAVI.
Limitations
This meta-analysis should be interpreted in the context of its limitations. While this is the largest meta-analysis to date to compare TTE to TEE for any PVL, we recognize that the sample size is still limited. Thus, our sample sizes for examining the outcomes of PVL grade may be underpowered to detect differences in outcomes. Only observational studies were found in the literature after a systematic literature review. Although there were no significant differences in baseline characteristics, including surgical risk scores, observational studies inherently include a risk of confounding variables. For example, PVL is influenced by many factors such as the type of prosthesis used, differences in procedural technique and center experience[29]. However, mode of anesthesia is the most concerning confounding variable. We acknowledge that differences in resource utilization may be the result of using MAC rather than substituting TTE for TEE alone. It is difficult to separate effects of anesthesia from the type of echocardiography as often the mode of anesthesia influences the choice of echocardiography performed. Furthermore, this study only analyzed early outcomes but did not assess late outcomes. Randomized clinical trials are needed to further investigate differences between TTE and TEE in TAVI.
In summary, the use of TTE-TAVI with MAC does not appear to adversely increase PVL rates compared to TEE-TAVI with GA. Furthermore, early mortality was similar between the two imaging modalities while resource utilization appeared to be lower with TTE, although this may have been associated with other factors such as mode of anesthesia.
Declarations
Authors’ contributionsData collection: Lam TK, Dixit AR
Data analysis/interpretation, drafting article: Lam TK
Approval of article: Lam TK, Tam DY, Dixit AR, Fremes SE
Concept/design: Tam DY, Fremes SE
Critical revision of article: Tam DY, Dixit AR, Fremes SE
Availability of data materialsNot applicable.
Financial support and sponsorshipDr. Derrick Tam is supported by a CIHR Fellowship. Dr. Stephen Fremes is supported in part by the Bernard S Goldman Chair.
Conflicts of interestAll authors declared that there are no conflicts of interest.
Ethical approval and consent to participateNot applicable.
Consent for publicationNot applicable.
Copyright© The Author(s) 2019.
REFERENCES
1. Mack MJ, Leon MB, Smith CR, Miller DC, Moses JW, et al. 5-year outcomes of transcatheter aortic valve replacement or surgical aortic valve replacement for high surgical risk patients with aortic stenosis (PARTNER 1): a randomised controlled trial. Lancet 2015;385:2477-84.
2. Smith CR, Leon MB, Mack MJ, Miller DC, Moses JW, et al. Transcatheter versus Surgical Aortic-Valve Replacement in High-Risk Patients. N Engl J Med 2011;364:2187-98.
3. Hamm CW, Möllmann H, Holzhey D, Beckmann A, Veit C, et al. The German Aortic Valve Registry (GARY): in-hospital outcome. Eur Heart J 2014;35:1588-98.
4. Sengupta PP, Wiley BM, Basnet S, Rajamanickman A, Kovacic JC, et al. Transthoracic echocardiography guidance for TAVR under monitored anesthesia care. JACC Cardiovasc Imaging 2015;8:379-80.
5. Xu B, Mottram PM, Lockwood S, Meredith IT. Imaging guidance for transcatheter aortic valve replacement: is transoesophageal echocardiography the gold standard? Hear Lung Circ 2017;26:1036-50.
6. Villablanca PA, Mohananey D, Nikolic K, Bangalore S, Slovut DP, et al. Comparison of local versus general anesthesia in patients undergoing transcatheter aortic valve replacement: a meta-analysis. Catheter Cardiovasc Interv 2018;91:330-42.
7. Hayek SS, Corrigan FE, Condado JF, Lin S, Howell S, et al. Paravalvular regurgitation after transcatheter aortic valve replacement: comparing transthoracic versus transesophageal echocardiographic guidance. J Am Soc Echocardiogr 2017;30:533-40.
8. Lerakis S, Hayek SS, Douglas PS. Paravalvular aortic leak after transcatheter aortic valve replacement: current knowledge. Circulation 2013;127:397-407.
9. Tam DY, Chu MW. Paravalvular leak after transcatheter aortic valve implantation: Trading one disease for another? Expert Rev Cardiovasc Ther 2014;12:407-11.
10. Guyatt GH, Oxman AD, Vist G, Kunz R, Brozek J, et al. GRADE guidelines: 4. rating the quality of evidence - study limitations (risk of bias). J Clin Epidemiol 2011;64:407-15.
11. Hozo SP, Djulbegovic B, Hozo I. Estimating the mean and variance from the median, range, and the size of a sample. BMC Med Res Methodol 2005;5:13.
12. Wan X, Wang W, Liu J, Tong T. Estimating the sample mean and standard deviation from the sample size, median, range and/or interquartile range. BMC Med Res Methodol 2014;14:135.
13. Higgins JPT, Thompson SG, Deeks JJ, Altman DG. Measuring inconsistency in meta-analyses. Br Med J 2003;327:557-60.
14. Gurevich S, Oestreich B, Kelly RF, Mbai M, Bertog S, et al. Outcomes of transcatheter aortic valve replacement using a minimalist approach. Cardiovasc Revascularization Med 2018;19:192-5.
15. Jabbar A, Khurana A, Mohammed A, Das R, Zaman A, et al. Local versus general anesthesia in transcatheter aortic valve replacement. Am J Cardiol 2016;118:1712-6.
16. Palermo C, Degnan M, Candiotti K, Salerno T, de Marchena E, et al. Monitored anesthesia care versus general anesthesia: experience with the medtronic CoreValve. J Cardiothorac Vasc Anesth 2016;30:1234-7.
17. Kiramijyan S, Ben-Dor I, Koifman E, Didier R, Magalhaes MA, et al. Comparison of clinical outcomes with the utilization of monitored anesthesia care vs. general anesthesia in patients undergoing transcatheter aortic valve replacement. Cardiovasc Revascularization Med 2016;17:384-90.
18. Attizzani GF, Alkhalil A, Padaliya B, Tam CC, Lopes JP, et al. Comparison of outcomes of transfemoral transcatheter aortic valve implantation using a minimally invasive versus conventional strategy. Am J Cardiol 2015;116:1731-6.
19. Dehédin B, Guinot PG, Ibrahim H, Allou N, Provenchère S, et al. Anesthesia and perioperative management of patients who undergo transfemoral transcatheter aortic valve implantation: an observational study of general versus local/regional anesthesia in 125 consecutive patients. J Cardiothorac Vasc Anesth 2011;25:1036-43.
20. Abud MA, Nau G, Candiello A, Padilla LT, Piccinini F, et al. Efficacy and safety of transfemoral transcatheter aortic valve replacement under general anesthesia versus local anesthesia with conscious sedation. Rev Argent Cardiol 2018;86:33-8.
21. Gauthier C, Momeni M, Astarci P, Baele P, Matta A, et al. Mid-term survival after transcatheter aortic valve implantation: results with respect to the anesthetic management and to the access route (transfemoral versus transapical). Ann Card Anaesth 2015;18:343-51.
22. Covello RD, Ruggeri L, Landoni G, Guarracino E, Bignami E, et al. Transcatheter implantation of an aortic valve: anesthesiological management. Minerva Anestesiol 2010;76:287-9.
23. Hosoba S, Yamamoto M, Shioda K, Sago M, Koyama Y, et al. Safety and efficacy of minimalist approach in transfemoral transcatheter aortic valve replacement: Insights from the optimized transcatheter valvular intervention-transcatheter aortic valve implantation (OCEAN-TAVI) registry. Interact Cardiovasc Thorac Surg 2018;26:420-4.
24. Miles LF, Joshi KR, Ogilvie EH, Densem CG, Klein AA, et al. General anaesthesia vs. Conscious sedation for transfemoral aortic valve implantation: a single UK centre before-and-after study. Anaesthesia 2016;71:892-900.
25. Condado JF, Haider MN, Lerakis S, Keegan P, Caughron H, et al. Does minimalist transfemoral transcatheter aortic valve replacement produce better survival in patients with severe chronic obstructive pulmonary disease? Catheter Cardiovasc Interv 2017;89:775-80.
26. Oguri A, Yamamoto M, Mouillet G, Gilard M, Laskar M, et al. Clinical outcomes and safety of transfemoral aortic valve implantation under general versus local anesthesia subanalysis of the French aortic national CoreValve and Edwards 2 registry. Circ Cardiovasc Interv 2014;7:602-10.
27. Sherifi I, Omar AMS, Varghese M, Weiner M, Anyanwu A, et al. Comparison of transesophageal and transthoracic echocardiography under moderate sedation for guiding Transcatheter Aortic Valve Replacement. Echo Res Pract 2014;7:602-10.
28. Sponga S, Perron J, Dagenais F, Mohammadi S, Baillot R, et al. Impact of residual regurgitation after aortic valve replacement. Eur J Cardio-thoracic Surg 2012;42:486-92.
Cite This Article
Export citation file: BibTeX | RIS
OAE Style
Lam TK, Tam DY, Dixit AR, Fremes SE. Transthoracic vs. transesophageal echocardiography in transcatheter aortic valve implantation: a systematic review and meta-analysis. Vessel Plus 2019;3:27. http://dx.doi.org/10.20517/2574-1209.2019.009
AMA Style
Lam TK, Tam DY, Dixit AR, Fremes SE. Transthoracic vs. transesophageal echocardiography in transcatheter aortic valve implantation: a systematic review and meta-analysis. Vessel Plus. 2019; 3: 27. http://dx.doi.org/10.20517/2574-1209.2019.009
Chicago/Turabian Style
Lam, Tiffany K., Derrick Y. Tam, Apurva R. Dixit, Stephen E. Fremes. 2019. "Transthoracic vs. transesophageal echocardiography in transcatheter aortic valve implantation: a systematic review and meta-analysis" Vessel Plus. 3: 27. http://dx.doi.org/10.20517/2574-1209.2019.009
ACS Style
Lam, TK.; Tam DY.; Dixit AR.; Fremes SE. Transthoracic vs. transesophageal echocardiography in transcatheter aortic valve implantation: a systematic review and meta-analysis. Vessel Plus. 2019, 3, 27. http://dx.doi.org/10.20517/2574-1209.2019.009
About This Article
Copyright
Data & Comments
Data

 Cite This Article 7 clicks
Cite This Article 7 clicks











Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at support@oaepublish.com.