Fractional flow reserve guided coronary artery bypass grafting - new developments and future perspectives
Abstract
The potential role of fractional flow reserve (FFR) in coronary artery bypass grafting (CABG) planning and post-CABG patency assessment are currently under intense investigation to determine whether the favourable outcomes reported with FFR-guided percutaneous coronary intervention can be translated to surgical practice. This review provides an overview of the principles that guide FFR measurement, the clinical evolution of FFR in CABG practice, the much anticipated outcomes of recent investigations that compare FFR-guided and angiography guided CABG and outlines the potential of alternative technology that may assist in ensuring ongoing improvement in surgical revascularization outcomes.
Keywords
Introduction
We are currently witnessing rapid evolution in diagnostic and interventional technology for coronary artery disease (CAD). It is now well recognized that the visual assessment of coronary artery stenosis by angiography[1] or intravascular ultrasound[2,3] do not accurately reflect its physiological impact on myocardial territory. Fractional flow reserve (FFR) emerged as a transcatheter tool to potentially quantify the physiological significance of coronary artery stenosis[4,5] and various reports over the last 2 decades repeatedly confirmed the favorable impact of FFR on percutaneous coronary intervention (PCI) decision-making and clinical outcomes[6-13]. These studies also suggested that less than 50% of angiographic significant lesions were functionally significant and that up to 25% of post-angiography guided-coronary artery bypass grafting (CABG) targets had no impact of physiological myocardial perfusion, potentially risking early graft failure, accelerate native artery disease and late graft failure exacerbation[14]. This review provides an outline of the principles that guide FFR measurement, the clinical evolution of FFR in CABG practice, the outcomes of recent investigations that compare FFR-guided and angiography guided CABG and describe potential alternative technology that may assist in ensuring ongoing improvement in surgical revascularization outcomes.
Fractional flow reserve as a physiological measure of coronary artery stenosis
FFR measurements usually form part of routine diagnostic radial or femoral access cardiac catheterization under local anaesthesia[6-13]. A pressure transducer within a fluoroscopically visible guide wire record the coronary arterial pressure distal to the identified lesion and once in position, facilitate the administration of a hyperemic stimulus by intracoronary or intravenous vasodilator (usually adenosine) injection. The mean arterial pressures from the pressure wire transducer and from the guide catheter are then used to calculate FFR, which is defined as the ratio of the average distal coronary pressure to average aortic pressure at maximal steady state hyperemia (normal value = 1.0). This equates to expressing the maximum achievable blood flow across an epicardial coronary stenosis to the maximum achievable blood flow in the absence of any stenosis. Various anatomical and clinical scenarios[15-24], which are outlined in Table 1, potentially influence FFR measurement accuracy that may result in deferral- or over-treatment of physiologically significant culprit lesions (FFR measurement ratio of 0.8 or less).
Potential contributing factors to FFR measurement inaccuracies
| Anatomical factor | Mechanism and Impact on FFR-measurement |
|---|---|
| Diffuse sequential lesions[5] | Multiple isolated sequential stenoses independently decrease coronary pressure and hyperaemic blood flow |
| Short left main stenosis[15,16] | Pressure damping and limited hyperaemic flow with optimal vasodilatation. FFR potentially overestimated |
| Acute coronary syndrome[17,18] | Cascade of coronary vascular receptors down-regulation, endothelial impairment and vasoconstriction. Potentially overestimate FFR/deleterious culprit-vessel deferral |
| Right heart failure[19] | Decrease coronary arterio-venous pressure gradients secondary to increases coronary venous- and microvascular pressures. FFR measurements potentially underestimated |
| Left heart failure[19] | Increased left ventricle end-diastolic pressure impedes myocardial perfusion. FFR increases 0.008 to 0.01/1 mmHg |
| Chronic multi-vessel disease collateralization[20,21] | Decrease in coronary artery-myocardial flow distribution. Microvascular disease is resistant to vasodilator hyperemia. FFR measurement potentially overestimated |
| Left ventricle outflow tract obstruction[22] | Left ventricle hypertrophy, elevated left ventricle end-diastolic pressure, increase microvascular resistance. FFR measurement potentially overestimated |
| Post-CABG conduit failure[23,24] | Competing flow, veno-arterial conduit resistance differences, arterial conduit autocrine activity, culprit-vessel pressure, collateral networks and sequential grafting techniques. FFR measurement inaccuracies due to technical challenges |
The evolution of FFR in coronary artery bypass grafting
The indications and procedural recommendation for CABG, which now include minimally invasive- and hybrid surgical procedures are well described in contemporary guidelines[25]. CABG of target lesions more than 50% stenosis were historically performed with the intention of restoring distal perfusion and to provide distal protection against native disease progression[26]. Subsets of early FFR-PCI measurement trials identified that up to 25% of target lesions had no myocardial perfusion improvement post-CABG with early graft failure, accelerate native artery disease and late graft failure exacerbation considered to be unfortunate consequences[14].
The well-defined benefits of CABG compared with angiography-guided PCI as reported in the ASCERT[27], SYNTAX[28,29], FREEDOM[30] and BEST trials[31] became subjected to intense scrutiny following the introduction of FFR and newer generation drug-eluting stent technology. Following the ground-breaking DEFER study[6], which established the basis for FFR-guided PCI investigation, the FAME[7,8] and FAME II trials[9,10] introduced the concept of physiological revascularization and reinforced the positive impact of FFR in PCI of multi-vessel disease, which included decreased repeat revascularization (4.3% in the FFR-guided group, 17.2% in angiography-guided group, P < 0.001), number of stents (mean 1.9 in FFR-guided group, mean 2.7 in angiography-guided group) and equivalence in 3-year comparative PCI-procedural costs ($16,792 for FFR-guided group, $16,737 for angiography guided group, P = 0.94). However, after 5 years, the authors reported no statistically significant difference in the incidence of major adverse cardiac events (MACE) between FFR-guided and angiography-guided PCI (31% in the angiography-guided group, 28% in the FFR-guided group, P = 0.31). A functional SYNTAX score of lesions with FFR less than 0.8 reclassified up to 32% of CABG candidates to lower risk groups treatable with both FFR-guided PCI and CABG as opposed to CABG alone[13]. Whether the favorable impact of FFR on PCI outcomes could be translated to surgical practice, became a subject of intense investigation.
The impact of FFR on CABG graft patency was investigated by Botman and coworkers[33], who reported a statistically significant 1 year graft occlusion incidence of 8.9% in FFR-guided vs. 21.4% of the angiography-guided CABG patients for both arterial (13.7% FFR-guided vs. 21.9% angiography-guided; P < 0.2) and venous (5.9% FFR-guided vs. 20.0% angiography-guided; P < 0.03) grafts. In those patients with angiographic stenosis of 50% to 70%, the graft patency was higher if the FFR was less than 0.75 and vessels diameter more than 2.0 mm.
A comparative study of angiography-guided- and FFR-guided CABG at the Cardiovascular Centre, Aalst (Belgium)[33] observed that FFR measurement resulted in a significant downgrade of multi-vessel disease functional severity, a subsequent decrease in the number of CABG grafts applied and no difference in the incidence of MACE between the 2 groups after 3-year follow-up. The incidence of severe recurrent angina was significantly lower in the FFR-guided CABG group (31% vs. 4%; P < 0.001). In a subgroup of 155 patients (25%) who underwent repeated coronary angiography for clinical indications, freedom from graft occlusion was higher in the FFR-guided group (21% in angiography-guided group, 5% in FFR-guided group, P = 0.031). The extended 6-year results were recently reported by Fournier and colleagues[34] and included 627 consecutive patients between 2006 and 2010. Both the rate of composite death or myocardial infarction (16% for FFR-guided group, 25% for angiography-guided group, P = 0.020) as well as death alone (11% for FFR-guided group, 18% for angiography-guided group, P = 0.013) were significantly lower in the FFR-guided CABG group. By Cox multivariate regression analysis, FFR-guidance was an independent predictor of reduced death or MI (P = 0.008). The Kaplan-Meier event rates diverged after 3 years to favour the FFR-guided CABG group. A propensity-matched cohort identified fewer MACE in the FFR-guided group (16% in FFR-guided group, 25% in angiographic-guided group, P < 0.02), which implies no increased risk of MACE by deferring FFR insignificant lesions.
The association of preoperative FFR on isolated total arterial CABG functionality 6 months postoperatively in patients with triple vessel disease were recently reported in the interim results of the IMPAG trial[35] as a 2-centre, single-arm, blinded study. The interim results of 63 patients (54 bilateral internal thoracic Y-graft configurations), included the evaluation of 199 arterial anastomoses, of which 135 were sequential anastomoses. Overall, 85% of the left internal thoracic artery (ITA) and 69% of the right ITA were functional and patent, which was statistically significantly associated with preoperative FFR values of 0.78. As arterial grafts are physiologically active and risk atrophy if subjected to competitive flow, the authors suggested that sequential anastomosis that provide continuous antegrade flow to multiple targets may provide graft protection for lesions in which FFR values were greater than 0.78. Antegrade flow in angiographic stenosed target vessels with large diameters, especially the right coronary artery (RCA), may still be adequate to cause significant competitive flow in arterial grafts and it is therefor suggested that the most appropriate FFR value for optimal RCA graft outcome was less than 0.71. The right ITA is often used for non-left anterior descending artery target vessels with poor distal run-off and apart from technical aspects, potentially explain the inferior graft patency.
New development and concerns of routinely applying fractional flow reserve-guided coronary artery bypass grafting
The recently published FARGO trial[36] evaluated graft patency and clinical outcome of 100 patients referred for CABG by a heart team after randomly being assigned to either FFR- or angiography-guided CABG. In FFR-guided CABG, coronary lesions with FFR > 0.80 were deferred, and a new graft plan was designed accordingly, whereas the surgeon was blinded to the FFR values in patients who underwent angiography-guided CABG. Angiographic follow-up at 6 months were available for 39 and 33 patients in the FFR- and angiography-guided groups respectively. Graft failures of all grafts, death, myocardial infarction, stroke and repeat revascularization were similar in both groups (16% vs. 12%; P = 0.97). After 6 months, deferred lesions (n = 24) showed a significant reduction in mean FFR from index to follow-up (0.89 ± 0.05 vs. 0.81 ± 0.11; P = 0.002). The authors concluded that FFR-guided CABG had similar graft failure rates and clinical outcomes as angiography-guided CABG. However, FFR was reduced significantly after 6 months in deferred lesions and may potentially result in adverse events over longer follow-up.
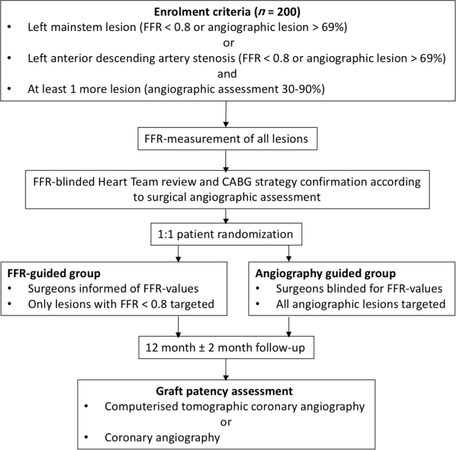
The GRAFFITI trial[37], of which the 12 month outcomes were presented at Euro-PCR in 2018 (Paris, France), was a prospective randomized trial that investigated the potential clinical benefits of FFR-guided vs. angiography-guided CABG in patients with left anterior descending or left mainstem disease and at least one other major coronary artery with angiographic intermediate stenosis (30%-90% diameter stenosis). The study design is described in Figure 1. The intended CABG strategy was based solely on coronary angiography after which patients underwent FFR- or angiography-guided randomization. In the FFR group, the surgical planning was revised according to the functional significance of each coronary stenosis after the FFR values were disclosed to the surgeons. After 12 months follow-up, the rate of graft occlusion (20% and 19% in angiography- and FFR-guided groups respectively, P = 0.885, 64.5% complete), rate of death (2% and 3% in angiography- and FFR-guided groups respectively, P = 0.65), myocardial infarction (2% and 0% in angiography- and FFR-guided groups respectively, P = 0.15), stroke (0% and 2% in angiography- and FFR-guided groups respectively, P = 0.16) and repeat revascularization (5% and 2% in angiography- and FFR-guided groups respectively, P = 0.35) were reported to be similar for both groups. Lesions with FFR measurement less than 0.8 were deferred in 53% and 29% in the angiography-guided and FFR-guided groups respectively, which suggested that FFR-guidance was associated with higher functional appropriateness (69% and 79% in angiography- and FFR-guided groups respectively). CABG was performed on 44% of stenotic lesions with preserved FFR and deferred on 53% of lesions with abnormal FFR, which translated to a significant reduced number of grafts.
Despite the paradigm shift toward physiological revascularization with all the benefits described, the value of complete anatomical revascularization remains relevant. Mulukutla and colleagues[38] recently reported a propensity-matched retrospective, observational analysis of patients with multi-vessel CAD who underwent angiography-guided CABG or FFR-PCI with second generation drug eluting stents between 2010 and 2018 and for whom data were available through the National Cardiovascular Data Registry or The Society of Thoracic Surgeons Adult Cardiac Surgery Database. Of the initial 6163 patients identified, the propensity-matched cohort included 844 in each group. The estimated 1-year mortality was 11.5% and 7.2% (P < 0.001) in the PCI and CABG groups respectively and overall MACE and individual outcomes of mortality, readmission, and repeat revascularization all favored CABG across all major clinical subgroups. CABG was angiography-guided and suggest that excellent outcomes that outperform PCI are achievable without FFR guidance.
The FUTURE trial, which was presented by Rioufol and colleagues at European Society of Cardiology Congress in Munich (Germany, 2018), was designed to explore the impact of FFR guided treatment strategies in patients with angiographic multi-vessel coronary disease in 31 French centers. In the FFR-guided group, FFR was performed on all target lesions, with FFR less than 0.80 regarded as eligible for PCI or CABG. There planned enrolment of 1,728 patients was halted due to an observed difference in all-cause mortality after 938 patients were randomized. The presenters highlighted three factors that could have played a role in the higher rate of mortality in the FFR-guided group: the lower-than-expected rate of CABG considering that all patients had multi-vessel disease, the higher rate of PCI in severe patients with a SYNTAX score over 32, and the high rate of ad hoc PCI (about 90% in both groups).
Potential alternative modalities that may improve coronary artery bypass grafting planning and decision-making
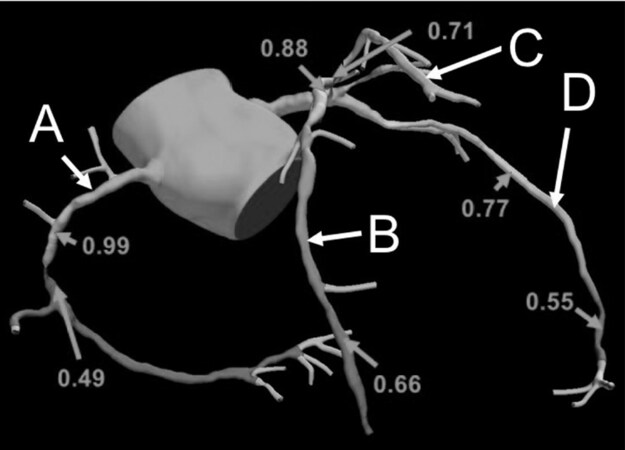
The rapid development in non-invasive imaging technology offers exciting potential alternatives to conventional invasive coronary angiography. Computerised tomographic coronary angiography (CTCA) is a non-invasive, repeatable, safe and efficient primary investigation in stable CAD that reduces the incidence of “negative” invasive angiography where no intervention is indicated or required[39]. Significant progress has been achieved regarding radiation exposure, with the introduction of dose-sparing protocols for ECG synchronization, the widespread use of lower tube voltages, and the development of iterative reconstruction algorithms. It has the major benefit of documented correlation with invasive FFR [Figure 2] and may result in a significant paradigm shift towards non-invasive coronary stenosis assessment and appropriate referral for treatment by CABG or PCI[40,41]. Nogaard and colleagues reported that 185 (98%) of 189 patients (mean age 59 years, 59% male) that underwent FFR-CTCA had conclusive results, with FFR-CTCA < 0.8 correlating with invasive coronary angiography in 73% of patients and 70% of vessels. In patients with FFR-CTCA lesions > 0.80, invasive coronary angiography was deferred with no adverse cardiac events observed during a median follow-up period of 12 months (range 6 to 18 months). They calculated per-patient sensitivity and specificity (95% CI) to identify myocardial ischemia as 86% and 79% for FFR-CTCA, 94% and 34% for standard CTCA and 64% and 83% for invasive coronary angiography respectively.
Figure 2. Computerised tomographic fraction flow reserve measurements of the right coronary (A), circumflex (B), intermediate (C), and left anterior descending artery (D)
Two non-hyperemic, invasive measures of inducing pressure might be useful for assessing the severity of coronary stenosis[42]. The resting distal coronary artery pressure/aortic pressure (Pd/Pa) is the ratio of distal coronary artery pressure to aortic pressure over the entire cardiac cycle. Instantaneous wave-free ratio (iFR) measures coronary pressure during a specific period of diastole when the resting resistance is the lowest[42]. Shiode and colleagues investigated the correlation between FFR-angiography, iFR and resting Pd/Pa by continuously measuring each component in 123 lesions in 103 patients with stable CAD by an intracoronary injection of papaverine. A receiver operator curve analysis revealed that the optimal iFR cut-off value for predicting an angiographic FFR of < 0.80 was 0.89 (sensitivity 84.1%, specificity 80.0%, diagnostic accuracy 81.3%), while the optimal resting Pd/Pa cut-off value was 0.92 (sensitivity 90.9%, specificity 78.5%, diagnostic accuracy 82.9%). Lesions with an iFR value < 0.89 and a Pd/Pa value < 0.92 were defined as double-positive lesions, while the lesions with an iFR value of > 0.89 and a Pd/Pa value of > 0.92 were defined as double-negative lesions. The ADVISE[43], ADVISE-e[44] and RESOLVE trials[45] also suggested that iFR compares favourably with FFR-angiography, may defer up to 16% of FFR significant lesions, may reduce procedural time, lower procedural cost, improve patient comfort and avoid side effects of adenosine, especially for patients at risk. The clinical application of iFR and Pd/Pa will soon be defined.
Conclusion
Various studies now confirm that physiological revascularization by FFR-guided CABG result in fewer target lesions and improved conduit patency in the short term. Even though intermediate follow-up results suggest no significant difference in clinical outcomes compared to angiography-guided CABG, the risk that angiographic significant/FFR insignificant lesions may progress to detrimental clinical events are of greatest concern. Current evidence therefor does not support the routine use of FFR in CABG planning. The current CABG procedure recommendation of complete angiographic- and total arterial revascularization with minimal aorta manipulation should remain the standard until future studies clarify the role of FFR in long term CABG outcomes[25]. Non-invasive CAD diagnostic modalities are rapidly developing and may offer exciting alternatives to FFR in planning CABG target lesions. The diagnostic-, shared decision-making-, informed consent- and therapeutic practices related to CAD treatment are sure to evolve with CABG to remain an invaluable- and excellent option.
Declarations
Authors’ contributionsRead and agreed to the manuscript as written: Van der Merwe J, Casselman F
Availability of data and materialsNot applicable.
Financial support and sponsorshipNone.
Conflicts of interestAll authors declared that there are no conflicts of interest.
Ethical approval and consent to participateNot applicable.
Consent for publicationNot applicable.
Copyright© The Author(s) 2019.
REFERENCES
1. Ahmadi A, Kini A, Narula J. Discordance between ischemia and stenosis, or PINSS and NIPSS: are we ready for new vocabulary? JACC Cardiovasc Imaging 2015;8:111-4.
2. Johnson NP, Kirkeeide RL, Gould KL. Coronary anatomy to predict physiology: fundamental limits. Circ Cardiovasc Imaging 2013;6:817-32.
3. Kern MJ, Samady H. Current concepts of integrated coronary physiology in the catheterization laboratory. J Am Coll Cardiol 2010;55:173-85.
4. Pijls NH, van Son JA, Kirkeeide RL, De Bruyne B, Gould KL. Experimental basis of determining maximum coronary, myocardial, and collateral blood flow by pressure measurements for assessing functional stenosis severity before and after percutaneous transluminal coronary angioplasty. Circulation 1993;87:1354-67.
5. Pijls NH, De Bruyne B, Peels K, Van Der Voort PH, Bonnier HJ, et al. Measurement of fractional flow reserve to assess the functional severity of coronary-artery stenoses. N Engl J Med 1996;334:1703-08.
6. Pijls NH, van Schaardenburgh P, Manoharan G, Boersma E, Bech JW, et al. Percutaneous coronary intervention of functionally nonsignificant stenosis: 5-year follow- up of the DEFER Study. J Am Coll Cardiol 2007;49:2105-11.
7. Tonino PA, De Bruyne B, Pijls NH, Siebert V, Ikeno F, et al. Fractional flow reserve vs. angiography for guiding percutaneous coronary intervention. N Engl J Med 2009;360:213-24.
8. Pijls NH, Fearon WF, Tonino PAO, Siebert U, Ikeno F, et al. Fractional flow reserve vs. angiography for guiding percutaneous coronary intervention in patients with multivessel coronary artery disease. J Am Coll Cardiol 2010;56:177-184.
9. De Bruyne B, Pijls NH, Kalesan B, Barbato E, Tonino PA, et al; FAME 2 Trial Investigators. Fractional flow reserve-guided PCI versus medical therapy in stable coronary disease. N Engl J Med 2012;367:991-1001.
10. De Bruyne B, Fearon WF, Pijls NH, Barbato E, Tonino P, et al; FAME 2 Trial Investigators. Fractional flow reserve-guided PCI for stable coronary artery disease. N Engl J Med 2014;371:1208-17.
11. Johnson NP, Tóth GG, Lai D, Zhu H, Açar G, et al. Prognostic value of fractional flow reserve: linking physiologic severity to clinical outcomes. J Am Coll Cardiol 2014;64:1641-54.
12. Mangiacapra F, Barbato E. From SYNTAX to FAME, a paradigm shift in revascularization strategies: the key role of fractional flow reserve in guiding myocardial revascularization. J Cardiovasc Med 2011;12:538-542.
13. Nam CW, Mangiacapra F, Entjes R, Chung IS, Sels JW, et al; FAME Study Investigators. Functional SYNTAX score for risk assessment in multivessel coronary artery disease. J Am Coll Cardiol 2011;58:1211-8.
14. Harskamp RE, Alexander JH, Ferguson TB, Hager R, Mack MJ, et al. Frequency and predictors of internal mammary artery graft failure and subsequent clinical outcomes: insights from the Project of Ex-vivo Vein Graft Engineering via Transfection (PREVENT) IV trial. Circulation 2016;133:131-8.
15. Toth GG, Johnson NP, Jeremias A, Pellicano M, Vranckx P, et al. Standardization of Fractional Flow Reserve Measurements. J Am Coll Cardiol 2016;68:742-53.
16. Kern MJ. My approach to the patient diagnosed with significant left main disease: Use of FFR, IVUS, and OCT. Trends Cardiovasc Med 2016;26:660-1.
17. Johnson NP, Gould KL, Di Carli MF, Taqueti VR. Invasive FFR and Noninvasive CFR in the Evaluation of Ischemia: What Is the Future? J Am Coll Cardiol 2016;67:2772-88.
18. Engstrom T, Kelbaek H, Helqvist S, Høfsten DE, Kløvgaard L, et al. Complete revascularisation versus treatment of the culprit lesion only in patients with ST-segment elevation myocardial infarction and multivessel disease (DANAMI-3- PRIMULTI): an open-label, randomised controlled trial. Lancet 2015;386:665-71.
19. Toth GG, De Bruyne B, Rusinaru D, Di Gioia G, Bartunek J, et al. Impact of Right Atrial Pressure on Fractional Flow Reserve Measurements: Comparison of Fractional Flow Reserve and Myocardial Fractional Flow Reserve in 1,600 Coronary Stenoses. JACC Cardiovasc Interv 2016;9:453-9.
20. van de Hoef TP, Nolte F, EchavarrIa-Pinto M, van Lavieren MA, Damman P, et al. Impact of hyperaemic microvascular resistance on fractional flow reserve measurements in patients with stable coronary artery disease: insights from combined stenosis and microvascular resistance assessment. Heart 2014;100:951-9.
21. Meuwissen M, Chamuleau SA, Siebes M, Schotborgh CE, Koch KT, et al. Role of variability in microvascular resistance on fractional flow reserve and coronary blood flow velocity reserve in intermediate coronary lesions. Circulation 2001;103:184-7.
22. Di Gioia G, Pellicano M, Toth GG, Casselman F, Adjedj J, et al. Fractional Flow Reserve-Guided Revascularization in Patients With Aortic Stenosis. Am J Cardiol 2016;117:1511-5.
23. Pellicano M, De Bruyne B, Toth GG, Casselman F, Wijns W, et al. Fractional flow reserve to guide and to assess coronary artery bypass grafting. Eur Heart J 2016;38:1959-68.
24. Aggarwal V, Stanislawski MA, Maddox TM, Nallamothu BK, Grunwald G, et al. Safety and effectiveness of drug-eluting versus bare-metal stents in saphenous vein bypass graft percutaneous coronary interventions: insights from the Veterans Affairs CART program. J Am Coll Cardiol 2014;64:1825-36.
25. Neumann FJ, Sousa-Uva M, Ahlsson A, Alfonso F, Banning AP, et al; ESC Scientific Document Group. 2018 ESC/EACTS Guidelines on myocardial revascularization. Eur Heart J 2019;40:87-165.
26. Dimitrova KR, Hoffman DM, Geller CM, Dincheva G, Ko W, et al. Arterial grafts protect the native coronary vessels from atherosclerotic disease progression. Ann Thorac Surg 2012;94:475-81.
27. Weintraub WS, Grau-Sepulveda MV, Weiss JM, O’Brien SM, Peterson ED, et al. Comparative effectiveness of revascularization strategies. N Engl J Med 2012;366:1467-76.
28. Serruys PW, Morice MC, Kappetein AP, Colombo A, Holmes DR, et al. Percutaneous coronary intervention versus coronary-artery bypass grafting for severe coronary artery disease. N Engl J Med 2009;360:961-72.
29. Mohr FW, Morice MC, Kappetein AP, Feldman TE, Ståhle E, et al. Coronary artery bypass graft surgery versus percutaneous coronary intervention in patients with three-vessel disease and left main coronary disease: 5-year follow-up of the randomised, clinical SYNTAX trial. Lancet 2013;381:629-38.
30. Farkouh ME, Domanski M, Sleeper LA, Siami FS, Dangas G, et al; FREEDOM Trial Investigators. Strategies for multivessel revascularization in patients with diabetes. N Engl J Med 2012;367:2375-84.
31. Park SJ, Ahn JM, Kim YH, Park DW, Yun SC, et al. Trial of everolimus-eluting stents or bypass surgery for coronary disease. N Engl J Med 2015;372:1204-12.
32. Botman CJ, Schonberger J, Koolen S, Penn O, Botman H, et al. Does stenosis severity of native vessels influence bypass graft patency? A prospective fractional flow reserve-guided study. Ann Thorac Surg 2007;83:2093-97.
33. Toth G, De Bruyne B, Casselman F, De Vroey F, Pyxaras S, et al. Fractional flow reserve-guided versus angiography-guided coronary artery bypass graft surgery. Circulation 2013;128:1405-11.
34. Fournier S, Toth GG, De Bruyne B, Johnson NP, Ciccarelli G, et al. Six-Year Follow-Up of Fractional Flow Reserve-Guided Versus Angiography-Guided Coronary Artery Bypass Graft Surgery. Circ Cardiovasc Interv 2018;11:e006368.
35. Glineur D, Grau JB, Etienne PY, Benedetto U, Fortier JH, Papadatos S, et al. Impact of preoperative fractional flow reserve on arterial bypass graft anastomotic function: the IMPAG trial. Eur Heart J 2019; doi: 10.1093/eurheartj/ehz329. [Epub ahead of print]
36. Thuesen AL, Riber LP, Veien KT, Christiansen EH, Jensen SE, et al. Fractional flow reserve versus angiographically-guided coronary artery bypass grafting. J Am Coll Cardiol 2018;72:2732-43.
37. Toth GG, De Bruyne B, Kala P, Ribichini FL, Casselman F, et al. Graft patency after FFR-guided versus angiography-guided coronary artery bypass grafting. The GRAFFITI trial. EuroIntervention 2019;aa-608 2019. doi: 10.4244/EIJ-D-19-00463.
38. Mulukutla SR, Gleason T, Sharbaugh M, Sultan I, Marroquin OC, et al. Coronary Bypass Versus Percutaneous Revascularization in Multivessel Coronary Artery Disease. Ann Thorac Surg 2019; doi: 10.1016/j.athoracsur.2019.02.064. [Epub ahead of print]
39. Norgaard BL, Hjort J, Gaur S, Hansson N, Bøtker HE, et al. Clinical Use of Coronary CTA-Derived FFR for Decision-Making in Stable CAD. JACC Cardiovasc Imaging 2017;10:541-50.
40. Curzen NP, Nolan J, Zaman AG, Nørgaard BL, Rajani R. Does the Routine Availability of CT-Derived FFR Influence Management of Patients With Stable Chest Pain Compared to CT Angiography Alone?: The FFRCT RIPCORD Study. JACC Cardiovasc Imaging 2016;9:1188-94.
41. Nørgaard BL, Leipsic J, Gaur S, Seneviratne S, Ko BS, et al. Diagnostic Performance of Noninvasive Fractional Flow Reserve Derived From Coronary Computed Tomography Angiography in Suspected Coronary Artery Disease. J Am Coll Cardiol 2014;63:1145-55.
42. Shiode N, Okimoto T, Tamekiyo H, Kawase T, Yamane K, et al. Comparison between the Instantaneous Wave-free Ratio and Resting Distal Coronary Artery Pressure/Aortic Pressure and the Fractional Flow Reserve: The Diagnostic Accuracy Can Be Improved by the Use of both Indices. Intern Med 2017;56:749-53.
43. Sen S, Escaned J, Malik IS, Mikhail GW, Foale RA, et al. Development and validation of a new adenosine-independent index of stenosis severity from coronary wave-intensity analysis: results of the ADVISE (ADenosine Vasodilator Independent Stenosis Evaluation) study. J Am Coll Cardiol 2012;59:1392-402.
44. Petraco R, Al-Lamee R, Gotberg M, Sharp A, Hellig F, et al. Real-time use of instantaneous wave-free ratio: results of the ADVISE in-practice: an international, multicenter evaluation of instantaneous wave-free ratio in clinical practice. Am Heart J 2014;168:739-48.
Cite This Article
Export citation file: BibTeX | RIS
OAE Style
Van der Merwe J, Casselman F. Fractional flow reserve guided coronary artery bypass grafting - new developments and future perspectives. Vessel Plus 2019;3:24. http://dx.doi.org/10.20517/2574-1209.2019.17
AMA Style
Van der Merwe J, Casselman F. Fractional flow reserve guided coronary artery bypass grafting - new developments and future perspectives. Vessel Plus. 2019; 3: 24. http://dx.doi.org/10.20517/2574-1209.2019.17
Chicago/Turabian Style
Van der Merwe, Johan, Filip Casselman. 2019. "Fractional flow reserve guided coronary artery bypass grafting - new developments and future perspectives" Vessel Plus. 3: 24. http://dx.doi.org/10.20517/2574-1209.2019.17
ACS Style
Van der, Merwe J.; Casselman F. Fractional flow reserve guided coronary artery bypass grafting - new developments and future perspectives. Vessel Plus. 2019, 3, 24. http://dx.doi.org/10.20517/2574-1209.2019.17
About This Article
Copyright
Data & Comments
Data
 Cite This Article 1 clicks
Cite This Article 1 clicks












Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at support@oaepublish.com.