Complications of transcatheter aortic valve replacement and rescue attempts
Abstract
As a novel treatment modality, transcatheter aortic valve replacement (TAVR) is widely used for patients with severe aortic valve stenosis who have high surgical risk worldwide. However, this promising alternative procedure has different types of complication risks including, cerebrovascular events, vascular complications, bleeding, coronary obstruction, myocardial infarction, valve regurgitation, valve malpositioning or migration, conduction disturbances and acute kidney injury which may occur during and/or after the procedure. These complications may be seen up to one third of the patients and some of them may need urgent surgical intervention and may have a higher risk of death. For preventing and overcoming these complications, pre-procedural evaluation of the patient by an effective “heart team” which consists of cardiologists, cardiac surgeons, radiologists and anesthesiologists in equal proportion is needed. Estimating the potential difficulties and complications, deciding the interventions to be performed in case of any complication may increase the success of the procedure and save the patients’ lives. In this article, we reviewed the possible complications of the TAVR procedure and described rescue procedures in case of complications, in the context of the literature.
Keywords
Introduction
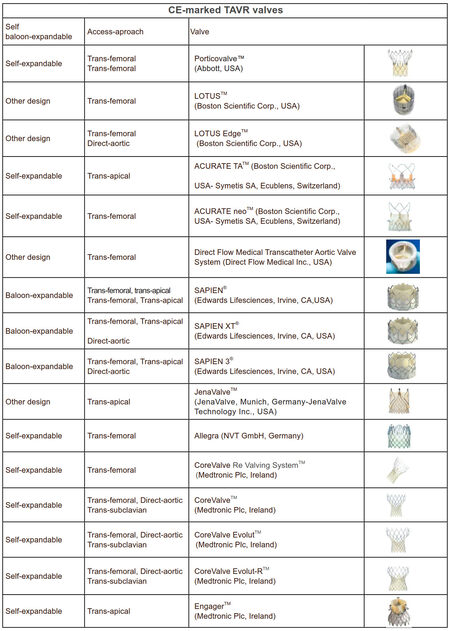
Transcatheter aortic valve replacement (TAVR) was firstly described as catheter-based implantation of a crimped valve, to the stenotic native aortic valve via transapical access-antegrade approach in 2002, as an alternative treatment method to surgical aortic valve replacement (SAVR) for patients who have high or prohibitive surgical risk[1,2]. Since 2002, TAVR is used increasingly all over the world. By the time, different access sites have been described and different TAVR valves have been developed [Figures 1 and 2].
Although its efficacy has been proven in patients with aortic valve stenosis having high surgical risks, as a less invasive catheterization procedure, it has varying types of complications that may increase morbidity, require urgent surgical intervention and even cause death. These complications can occur anytime during and/or after the procedure, and include cerebrovascular events, vascular complications, bleeding, coronary obstruction, myocardial infarction, valve regurgitation, valve malpositioning or migration, conduction disturbances and acute kidney injury. With the advances in medical equipment and systems, improvements in procedural techniques together with increasing experience and advances in patients’ imaging, these procedural complications decreased dramatically. However, if occur, complications still remain the major factors affecting the success of the procedure. To prevent and/or overcome these complications, all TAVR patients should be evaluated by the “heart team” which consists of cardiologists, cardiac surgeons, radiologists and anesthesiologists in equal proportion. The risks and/or difficulties of anesthesia and SAVR procedure should be put forth by the cardiac surgeons and anesthesiologists, and declared to the patient. Once the decision of performing TAVR procedure has been taken, structures and calcification loads of the aortic valve, aortic annulus, aorta and access vessels should be evaluated by cardiologists, cardiac surgeons and radiologists via CT images, angiogram and echocardiographic findings. The TAVR valve planned to be used, potential difficulties of the procedure and possible complications should be determined and in case of complications, rescue attempts should be planned before the procedure by the “heart team”. The procedure should be performed in a hybrid operating room and surgical backup should be available whenever needed.
However, in all centers where TAVR is performed, it seems that a heart team with equal participation of specialists is not established and managed. Performing the procedure under this inappropriate condition may cause doctors to inform the procedure as a risk-free intervention to the patients, to get out of the TAVR indications such as performing the procedure according to the patient’s wish only and to be caught unprepared against the complications.
Here, for highlighting pre-procedural evaluation of the patients and being prepared against the complications of TAVR, we reviewed the possible complications of the TAVR procedure and described rescue procedures and/or treatment options in case of complications, in the context of the literature.
Vascular complications
The vascular complications of TAVR may be evaluated under two subheadings.
Minor/major vascular complications
Minor Vascular Complications
Vascular access injuries, those do not cause tissue malperfusion and do not require surgery[3].
Major Vascular Complications
All other vascular injuries, those cause tissue malperfusion, require blood transfusion over 4 units or surgery.
Heart team has the key role in preventing and/or overcoming major vascular complications. Not only the status of aortic valve and device landing zone, a full evaluation including the status of the access-site, access artery diameter, its stenosis and/or calcification, sharp angulations and/or tortuosity of the conducting arteries, should be done by using computerized tomography and catheter angiography.
Vascular access-site/device landing zone complications
Vascular Access-Site Complications
Vascular access-site complications are mainly caused by the mismatch of access artery and sheaths of delivery system. Sex (female), calcification status of the access artery, ratio of the sheath to access artery diameter (> 1.05)[4] and the experience of the operator were determined as major predictors of vascular access-site complications[5,6]. With the improvements in the delivery systems (decreased diameters), improvements in the pre-procedural patient evaluation and increased surgical experience, the vascular access-site complications decreased nowadays[7]. Despite all the improvements, if the conducting arteries have sharp angulations, tortuosity or untreated aneurysms, and the conducting artery lumen is narrower than 6 mm with calcifications, the trans-femoral, trans-subclavian and trans-carotid accesses are not recommended, instead, trans-apical or direct-aortic accesses should be used.
In case of any complication, angiographic evaluation of the artery may be the urgent diagnosis method and an acute hypotension without other causes may also support the diagnosis of major arterial injury. Urgent endovascular or surgical repair is recommended for treatment.
Device landing zone complications
Rupture of the device landing-zone is a rarely encountered complication (1%), but has a high mortality risk (48%-50%)[8,9]. The presence of severe annular, sub-annular, left ventricular outflow tract calcifications and valve over sizing were determined as the predictors of this complication[10]. From the perspective of tissue quality, patients older than 90 years, chronic steroid users and immunosuppressed hosts have a higher risk of annular injury. Device landing-zone complications such as injury, rupture or dissection of aorta, ventricular septal defect and aorto-ventricular fistula are mostly seen in implantation of balloon-expandable valves or in balloon dilatation of a self-expandable valve after implantation[11]. Smaller annular area (< 300 cm2) may increase the annular rupture due to relative valve oversizing[12]. Also, aggressive oversizing of the prosthesis, may decrease significant aortic regurgitation but induce conduction disorders requiring pacemaker implantations[13].
In case of complication, trans-esophageal echocardiography may give critical information about new pericardial effusion or tamponade, aortic root injury and aortic dissection. The occurrence of an acute hypotension supports the diagnosis. If the problem is only aortic root hematoma with no rupture, hemodynamic support with inotropes, reversal of anticoagulation, then transfusion of fresh frozen plasma and close observation may be enough. Otherwise, if there is rupture, cardiac tamponade occurs frequently and reversal of anticoagulation, pericardial drainage or surgical repair are recommended[14].
Aortic valve regurgitation
Aortic valve regurgitation is frequently seen after TAVR and can be evaluated under two subheadings.
Paravalvular leak
The incidence of paravalvular leak is 50%-85%. Whilst most of them are mild, moderate and/or severe leaks are seen up to 24%[15] that increase the mortality of the procedure up to 4 times in the first year[16,17]. Occurrence of paravalvular leaks can be explained by 3 mechanisms: (1) prosthetic valve-annulus size mismatch; (2) inappropriate placement of the prosthetic valve; and (3) incomplete apposition of the stent due to deformed native structure.
Aortic root calcification, its degree and geometric distribution are the main factors affecting the native structure. Asymmetric and severe calcifications may deform the prosthesis resulting in paravalvular leaks. Assessing the aortic root calcification with echocardiographic examination and/or Agatston score, may decrease the risk of paravalvular leak[18].
The use of self-expandable valves is a major determinant for significant paravalvular leak. The studies have shown that self-expandable valves were associated with moderate-severe paravalvular leak compared with balloon-expandable valves (19.8% vs. 12.2%)[19].
Central leak
The incidence of moderate or severe central leak is 4.5%-11.7%[20] and usually occurs due to structural dysfunction of the valve. Central leak can be the result of leaflet restriction or damage, during crimping or implantation as well as over dilatation of the valve[21]. Post implantation dilatation of the prosthetic valve may also cause central leak[22].
In case of any complication, aortic root angiography is performed for the quantification of central leak. Intra-procedural echocardiography may be performed for determining the severity of leak and the location of the prosthetic valve. Increase of left ventricular end-diastolic pressure and decrease of aortic diastolic blood pressure also support the diagnosis. If the leak is central, gentle probing of leaflets with a soft wire and/or catheter or delivery of a second prosthetic valve may solve the problem. The management of paravalvular leaks is controversial. Mild degrees may be clinically followed as they are thought to be not progressive. However, more severe degrees of leaks may deserve intervention. Usually, balloon post-dilatation is the first option, using a slightly oversized balloon.
Repositioning of the implanted prosthetic valve, delivery of a second prosthetic valve and percutaneus vascular occlusion devices may be the other choices for the treatment. However, in large and high volume leaks, for implanting the appropriate device, large sheaths may be needed. Particularly in self-expanding prostheses, valve struts and calcification of the annulus may complicate the advancement of delivery systems mainly when using large sheaths[23].
Otherwise, SAVR should be performed for both types of leaks[14].
Prosthetic valve malpositioning
Valve malpositioning usually occurs during or just after valve implantation. However, rare delayed migration cases together with acute heart failure and/or cardiogenic shock have been reported in literature[24]. The incidence of the prosthetic valve malpositioning is about 1.3% (CoreValve® 2.3% vs. Edwards SAPIEN® valve 1.0%[20]. The predisposing factors for the prosthetic valve malpositioning can be listed as: (1) incorrect assessment of the aortic annulus; (2) incorrect implantation of the prosthetic valve; (3) insufficient or early termination of rapid ventricular pacing; (4) presence of prosthetic mitral valve; and (5) presence of severe mitral annular calcification extending to anterior leaflet and left ventricular outflow tract.
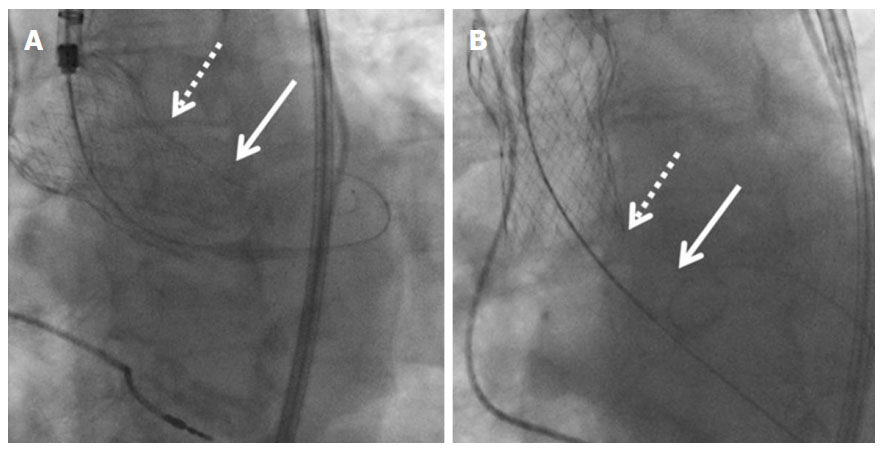
In case of any complication, aortography and trans-esophageal echocardiography are performed for evaluating the position and confirming the malposition or migration of prosthetic valve [Figure 3A and B].
Figure 3. A: An angiographic view of CoreValve® prosthesis before final delivery; B: final position of prosthesis in ascending aorta before emergent surgery (solid arrows show the annulus of the native aortic valve and dotted arrows show the sino-tubular junction)
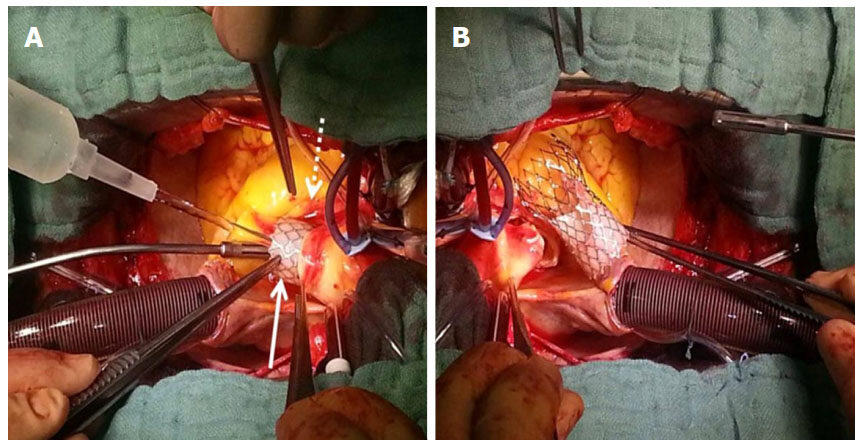
Hemodynamic status of the patient, final position and the type of prosthetic valve determine the treatment. For self-expandable ones, if the prosthetic valve is still attached to the delivery system, it may be re-captured or deployed to descending aorta. If not, it may be snared in the aortic direction or a second prosthetic valve may be implanted as valve-in-valve. For migration of the balloon expandable ones, prosthetic valve may be pulled to descending aorta via an inflated balloon inside. In case of unsuccessful bailout maneuvers, urgent surgical removal of the prosthetic valve and SAVR should be performed[14][Figure 4A and B].
Figure 4. A: An intraoperative view of migrated CoreValve® prosthesis (solid arrow) through aortic incision at the level of sino-tubular junction (dotted arrow). The surgical field is flushed with cold saline solution to soften the rigid nitinol struts of the prosthetic aortic valve for smooth extraction through aortotomy; B: an intraoperative view of migrated Medtronic-CoreValve® prosthesis removed from ascending aorta
Coronary obstruction
The incidence of coronary obstruction is about 0.8% for the procedures which are performed to native aortic valve and 3.5% for the procedures which are performed to degenerative bioprosthetic aortic valve[25,26]. The risk factors for coronary obstruction may be listed as: (1) low coronary ostium height (< 12 mm); (2) narrow sinus valsalva; (3) small sinotubuler junction; (4) low sinus valsalva height (< 30 mm); (5) bulky calcification of the aortic valve leaflets; and (6) oversized prosthetic valve.
Closure of the coronary ostium by the calcific aortic valve leaflets is the most encountered cause of the coronary obstruction[27] and also reported to be more frequent in women and in patients with prior surgical bioprosthesis. In the CHOICE trial, two patients belonging to the balloon-expandable valve group had coronary obstruction as opposed to none in the self-expandable valve group[28].
In case of complication; coronary obstruction manifests itself with acute hypotension, segment (ST) elevation, ventricular arrhythmias and/or cardiac arrest. Because of the high hemodynamic collapse risk, an emergent aortography or selective angiography to the obstructed coronary artery with stent implantation should be performed. The patient may be placed on mechanical circulatory support for allowing the operators to gain time for intervention. Failure of percutaneous coronary intervention indicates the necessity of a coronary bypass grafting operation for the treatment of this complication.
Myocardial infarction
The incidence of peri-procedural myocardial infarction is about 1.1% (in transapical approach 1.9% vs. in trans-arterial approach 0.8%)[8,20]. The reasons of peri-procedural myocardial infarction can be listed as[29]: (1) myocardial ischemia due to rapid ventricular pacing; (2) myocardial ischemia due to hypotension; (3) microembolisms to coronary arteries; (4) compression of the myocardium due to expansion of the prosthetic valve; 5. trauma to the ventricular apex in the trans-apical approach.
Presence of chest pain and/or shortness of breath, ST changes, pathological Q wave, hemodynamic instability, ventricular arrhythmia, new or worsened heart failure, elevated levels of cardiac biochemical markers (particularly CK-MB) in the post-procedural 72 h, detection of the loss of viable myocardium on imaging and ventricular wall motion abnormality also indicate the peri-procedural myocardial infarction[3].
In case of such complications, selective coronary angiography and percutaneous coronary interventions should be performed. According to the results, medical treatment and/or coronary artery bypass grafting operation may be the treatment options.
Cerebrovascular complications
The incidence of strokes and transient ischemic attacks in a month after TAVR procedure vary between 3%-7%[30,31]. The majority of these cerebrovascular complications (50%-70%) are seen in the first 24 h after the procedure and neither the type of prosthetic valve, nor the access route has any effect over incidence of cerebrovascular complications[20]. After the TAVR procedure, in one third of the patients, new onset atrial fibrillation may be encountered. The cerebrovascular complications that occur after the first 24 h are thought to be related with this new onset atrial fibrillation[32]. Studies revealed that the origin of embolic material was usually native aortic valve leaflets or aortic wall[33]. Thus, avoiding frequent aortic balloon dilatation and limiting the manipulations of large catheters in the aortic arch, were suggested to reduce the cerebrovascular complications[34].
In case of complication, in large ischemic cerebrovascular events, mechanical retrieval of the embolic material via catheter may be performed. Otherwise, conservative treatment should be performed[14]. Antiplatelet and anticoagulant agents should be used during and after the procedure. In the presence of newly onset atrial fibrillation, anti-arrhythmic drugs should also be added to the treatment.
Bleeding
Life-threatening bleeding
Occurrence in critical areas, development of severe hypotension or shock, decrease of hemoglobin value more than 5 g/dL or requirement of red blood cells transfusion more than 4 units, indicate the life-threatening bleeding.
Major bleeding
Bleedings that do not meet the life-threatening bleeding criteria but cause the decrease of hemoglobin value equal to or more than 3 g/dL and the ones those require 2-3 units of red blood cells transfusion can be defined as major bleeding.
Minor bleeding
All bleedings other than life-threatening and major bleedings can be described as minor bleeding.
Cardiac tamponade due to bleeding to the pericardium is seen in about 3%-4% of the patients who underwent TAVR and causes high rate of death (24%)[35]. Of the access-site complications, 69% is bleeding and 23%-31% of them are life-threatening ones. Digestive tract, the retro-peritoneum, and the pleura may be listed as the other sources of bleedings.
In case of such complications, the anticoagulation should be reversed and if needed transfusion of fresh frozen plasma and/or red blood cells should be performed. Hemodynamic conditions and hemoglobin levels should be stabilized. If feasible, the source of the bleeding should be treated surgically.
Cardiac conduction abnormalities
Conduction system damages are one of the major complications of TAVR and can be listed as: (1) prolonged atrio-ventricular (AV) conduction time; (2) AV block; (3) left bundle branch block; and (4) need for permanent pacemaker implantation.
The thickness of the ventricular septum, thickness of the non-coronary aortic cusp, implantation depth of the prosthetic valve in the left ventricular outflow tract, post implantation dilatation of the prosthetic valve, type of prosthetic valve and pre-existence of right bundle branch block can be listed as the risk factors for occurrence of conduction abnormalities[36,37]. The incidence of conduction abnormalities after TAVR varies between 5.7%-42.5%[38]. The incidence of AV block varies between 24.5%-25.8% for CoreValve® and 5.9%-6.5% for Edwards SAPIEN® valve[39]. Besides the prosthetic valve, manipulation of the guide wires and catheter systems in the left ventricular outflow tract may also cause temporary or permanent conduction system injuries. Most of the conduction abnormalities occur during the procedure (after the isolated aortic balloon valvuloplasty and before the implantation of the prosthetic valve)[40]. New left bundle branch block is the most seen conduction abnormality with the rate of 25%-85% for CoreValve® and 8%-30% for Edwards SAPIEN® valve[41]. The risk of AV block is higher for CoreValve® due to its self-expandable design and the possible deeper implantation into the left ventricular outflow tract. For preventing the complications related to conduction pathways, patients should be carefully screened for risk factors.
In case of such complications, trans-venous pacemaker implantation with conversion to permanent pacemaker is the most common treatment option[14].
Acute renal injury
The incidence of acute renal injuries after TAVR is about 22% and less than half of them are acute renal injuries in stage 2 or stage 3 (8.4%)[42]. The predisposing factors for acute renal injuries can be listed as: (1) chronic renal disease; (2) peripheral vascular disease; (3) diabetes mellitus; (4) hypoperfusion during rapid ventricular pacing; and (5) aortic plaque embolism in the renal arteries.
In case of any renal complication, the cessation of nephrotoxic drugs and the start of hydration procedure should be performed. If needed hemodialysis may be the treatment option.
Death
The mortality incidence after TAVR varies between 5%-10%. No significant difference about mortality has been reported between the self-expandable and balloon expandable prosthetic valve implantation[2,8]. However, significant difference is present between the trans-apical and trans-arterial implantation of the balloon expandable prosthetic valve[20]. The cause of death is mostly originated from the heart (75%) and occurs in the first 48 h after the procedure. After the first 48 h, non-cardiac reasons are the most encountered ones with an incidence of 69%[43]. Whilst heart failure, cardiac tamponade and arrhythmias are the most seen cardiac reasons; infection, sepsis and stroke are the most seen non-cardiac reasons of death.
Conclusion
TAVR procedure is increasingly used all over the world each day. Despite all procedural improvements and technical advances, TAVR procedure still has severe complication risks. It seems that the most important point of preventing and/or overcoming these complications is having an effective heart team. A good patient evaluation by each member of the team, appropriate patient selection, determining the procedural difficulties before the procedure may reduce the complications. Being prepared against the complications, may allow the most needed time to perform the rescue attempts and save the patients’ lives.
Declarations
Authors’ contributionsDesign: Depboylu B
Literature research, data analysis, manuscript writing: Depboylu B, Yazman S
Manuscript editing, manuscript revision: Harmandar B
Availability of data and materialsNot applicable.
Financial support and sponsorshipNone.
Conflicts of interestAll authors declared that there are no conflicts of interest.
Ethical approval and consent to participateNot applicable.
Consent for publicationNot applicable.
Copyright© The Author(s) 2018.
REFERENCES
1. Cribier A, Eltchaninoff H, Bash A, Borenstein N, Tron C, Bauer F, Derumeaux G, Anselme F, Laborde F, Leon MB. Percutaneous transcatheter implantation of an aortic valve prosthesis for calcific aortic stenosis: first human case description. Circulation 2002;106:3006-8.
2. Smith CR, Leon MB, Mack MJ, Miller DC, Moses JW, Svensson LG, Tuzcu EM, Webb JG, Fontana GP, Makkar RR, Williams M, Dewey T, Kapadia S, Babaliaros V, Thourani VH, Corso P, Pichard AD, Bavaria JE, Herrmann HC, Akin JJ, Anderson WN, Wang D, Pocock SJ; PARTNER Trial Investigators. Transcatheter versus surgical aortic-valve replacement in high-risk patients. N Engl J Med 2011;364:2187-98.
3. Leon MB, Piazza N, Nikolsky E, Blackstone EH, Cutlip DE, Kappetein AP, Krucoff MW, Mack M, Mehran R, Miller C, Morel MA, Petersen J, Popma JJ, Takkenberg JJ, Vahanian A, van Es GA, Vranckx P, Webb JG, Windecker S, Serruys PW. Standardized endpoint definitions for transcatheter aortic valve implantation clinical trials: a consensus report from the Valve Academic Research Consortium. Eur Heart J 2011;32:205-17.
4. Chaudhry MA, Sardar MR. Vascular complications of transcatheter aortic valve replacement: a concise literature review. World J Cardiol 2017;9:574-82.
5. Généreux P, Webb JG, Svensson LG, Kodali SK, Satler LF, Fearon WF, Davidson CJ, Eisenhauer AC, Makkar RR, Bergman GW, Babaliaros V, Bavaria JE, Velazquez OC, Williams MR, Hueter I, Xu K, Leon MB; PARTNER Trial Investigators. Vascular complications after transcatheter aortic valve replacement: insights from the PARTNER (Placement of AoRTicTraNscathetER Valve) trial. J Am Coll Cardiol 2012;60:1043-52.
6. Van Mieghem NM, Chieffo A, Dumonteil N, Tchetche D, van der Boon RM, Buchanan GL, Marcheix B, Vahdat O, Serruys PW, Fajadet J, Carrié D, Colombo A, de Jaegere PP. Trends in outcome after transfemoral transcatheter aortic valve implantation. Am Heart J 2013;165:183-92.
7. Hayashida K, Lefèvre T, Chevalier B, Hovasse T, Romano M, Garot P, Mylotte D, Uribe J, Farge A, Donzeau-Gouge P, Bouvier E, Cormier B, Morice MC. True percutaneous approach for transfemoral aortic valve implantation using the Prostar XL device: impact of learning curve on vascular complications. JACC Cardiovasc Interv 2012;5:207-14.
8. Généreux P, Head SJ, Van Mieghem NM, Kodali S, Kirtane AJ, Xu K, Smith C, Serruys PW, Kappetein AP, Leon MB. Clinical outcomes after transcatheter aortic valve replacement using valve academic research consortium definitions: a weighted meta-analysis of 3,519 patients from 16 studies. J Am Coll Cardiol 2012;59:2317-26.
9. Barbanti M, Yang TH, Rodès Cabau J, Tamburino C, Wood DA, Jilaihawi H, Blanke P, Makkar RR, Latib A, Colombo A, Tarantini G, Raju R, Binder RK, Nguyen G, Freeman M, Ribeiro HB, Kapadia S, Min J, Feuchtner G, Gurtvich R, Alqoofi F, Pelletier M, Ussia GP, Napodano M, de Brito FS Jr, Kodali S, Norgaard BL, Hansson NC, Pache G, Canovas SJ, Zhang H, Leon MB, Webb JG, Leipsic J. Anatomical and procedural features associated with aortic root rupture during balloon-expandable transcatheter aortic valve replacement. Circulation 2013;128:244-53.
10. Blanke P, Reinöhl J, Schlensak C, Siepe M, Pache G, Euringer W, Geibel-Zehender A, Bode C, Langer M, Beyersdorf F, Zehender M. Prosthesis oversizing in balloon-expandable transcatheter aortic valve implantation is associated with contained rupture of the aortic root. Circ Cardiovasc Interv 2012;5:540-8.
11. Revilla Martínez MI, Gutiérrez García H, San Román Calvar JA. Interventricular septum rupture after transcatheter aortic valve implantation. Eur Heart J 2012;33:190.
12. Nakashima M, Watanabe Y. Transcatheter aortic valve ımplantation in small anatomy: patient selection and technical challenges. Interv Cardiol 2018;13:66-8.
13. Debry N, Sudre A, Elquodeimat I, Delhaye C, Schurtz G, Bical A, Koussa M, Fattouch K, Modine T. Prognostic value of the ratio between prosthesis area and indexed annulus area measured by MultiSlice-CT for transcatheter aortic valve implantation procedures. J Geriatr Cardiol 2016;13:483-8.
14. Otto CM, Kumbhani DJ, Alexander KP, Calhoon JH, Desai MY, Kaul S, Lee JC, Ruiz CE, Vassileva CM. 2017 ACC expert consensus decision pathway for transcatheter aortic valve replacement in the management of adults with aortic stenosis: a report of the american college of cardiology task force on clinical expert consensus documents. J Am Coll Cardiol 2017;69:1313-46.
15. Lerakis S, Hayek SS, Douglas PS. Paravalvular aortic leak after transcatheter aortic valve replacement: current knowledge. Circulation 2013;127:397-407.
16. Athappan G, Patvardhan E, Tuzcu EM, Svensson LG, Lemos PA, Fraccaro C, Tarantini G, Sinning JM, Nickenig G, Capodanno D, Tamburino C, Latib A, Colombo A, Kapadia SR. Incidence, predictors, and outcomes of aortic regurgitation after transcatheter aortic valve replacement: meta-analysis and systematic review of literature. J Am Coll Cardiol 2013;61:1585-95.
17. Sinning JM, Vasa-Nicotera M, Chin D, Hammerstingl C, Ghanem A, Bence J, Kovac J, Grube E, Nickenig G, Werner N. Evaluation and management of paravalvular aortic regurgitation after transcatheter aortic valve replacement. J Am Coll Cardiol 2013;62:11-20.
18. Ryś M, Hryniewiecki T, Michałowska I, Stokłosa P, Różewicz-Juraszek M, Chmielak Z, Dąbrowski M, Mirota K, Szymański P. Quantitative estimation of aortic valve calcification in multislice computed tomography in predicting the development of paravalvular leaks following transcatheter aortic valve replacement. Postepy Kardiol Interwencyjnej 2018;14:85-9.
19. Généreux P, Head SJ, Hahn R, Daneault B, Kodali S, Williams MR, van Mieghem NM, Alu MC, Serruys PW, Kappetein AP, Leon MB. Paravalvular leak after transcatheter aortic valve replacement: the new Achilles' heel? A comprehensive review of the literature. J Am Coll Cardiol 2013;61:1125-36.
20. Khatri PJ, Webb JG, Rodés-Cabau J, Fremes SE, Ruel M, Lau K, Guo H, Wijeysundera HC, Ko DT. Adverse effects associated with transcatheter aortic valve implantation: a meta-analysis of contemporary studies. Ann Intern Med 2013;158:35-46.
21. Holmes DR Jr, Mack MJ, Kaul S, Agnihotri A, Alexander KP, Bailey SR, Calhoon JH, Carabello BA, Desai MY, Edwards FH, Francis GS, Gardner TJ, Kappetein AP, Linderbaum JA, Mukherjee C, Mukherjee D, Otto CM, Ruiz CE, Sacco RL, Smith D, Thomas JD. 2012 ACCF/AATS/SCAI/STS expert consensus document on transcatheter aortic valve replacement. J Am Coll Cardiol 2012;59:1200-54.
22. Al-Attar N, Himbert D, Vahanian A, Nataf P. Severe intraprosthetic regurgitation by immobile leaflet after trans-catheter aortic valve implantation. Eur J Cardiothorac Surg 2011;39:591-2.
23. Estévez-Loureiro R, Benito-González T, Gualis J, Pérez de Prado A, Cuellas C, Fernandez-Vazquez F. Percutaneous paravalvular leak closure after CoreValve transcatheter aortic valve implantation using an arterio-arterial loop. J Thorac Dis 2017;9:E103-8.
24. Pang PY, Chiam PT, Chua YL, Sin YK. A survivor of late prosthesis migration and rotation following percutaneous transcatheter aortic valve implantation. Eur J Cardiothorac Surg 2012;41:1195-6.
25. Dvir D, Webb J, Brecker S, Bleiziffer S, Hildick-Smith D, Colombo A, Descoutures F, Hengstenberg C, Moat NE, Bekeredjian R, Napodano M, Testa L, Lefevre T, Guetta V, Nissen H, Hernández JM, Roy D, Teles RC, Segev A, Dumonteil N, Fiorina C, Gotzmann M, Tchetche D, Abdel-Wahab M, De Marco F, Baumbach A, Laborde JC, Kornowski R. Transcatheter aortic valve replacement for degenerative bioprosthetic surgical valves: results from the global valve-in valve registry. Circulation 2012;126:2335-44.
26. Paradis JM, Del Trigo M, Puri R, Rodés-Cabau J. Transcatheter valve-in-valve and valve-in-ring for treating aortic and mitral surgical prosthetic dysfunction. J Am Coll Cardiol 2015;66:2019-37.
27. Ribeiro HB, Nombela-Franco L, Urena M, Mok M, Pasian S, Doyle D, DeLarochellière R, Côté M, Laflamme L, DeLarochellière H, Allende R, Dumont E, Rodés-Cabau J. Coronary obstruction following transcatheter aortic valve implantation: a systematic review. JACC Cardiovasc Interv 2013;6:452-61.
28. Abdel-Wahab M, Mehilli J, Frerker C, Neumann FJ, Kurz T, Tölg R, Zachow D, Guerra E, Massberg S, Schäfer U, El-Mawardy M, Richardt G; CHOICE investigators. Comparison of balloon-expandable versus self-expandable valves in patients undergoing transcatheter aortic valve replacement: the CHOICE randomized clinical trial. JAMA 2014;311:1503-14.
29. Rodés-Cabau J, Gutiérrez M, Bagur R, De Larochellière R, Doyle D, Côté M, Villeneuve J, Bertrand OF, Larose E, Manazzoni J, Pibarot P, Dumont E. Incidence, predictive factors, and prognostic value of myocardial injury following uncomplicated transcatheter aortic valve implantation. J Am Coll Cardiol 2011;57:1988-99.
30. Miller DC, Blackstone EH, Mack MJ, Svensson LG, Kodali SK, Kapadia S, Rajeswaran J, Anderson WN, Moses JW, Tuzcu EM, Webb JG, Leon MB, Smith CR; PARTNER Trial Investigators and Patients. Transcatheter (TAVR) versus surgical (AVR) aortic valve replacement: occurrence, hazard, risk factors, and consequences of neurologic events in the PARTNER trial. J Thorac Cardiovasc Surg 2012;143:832-43.
31. Stortecky S, Windecker S, Pilgrim T, Heg D, Buellesfeld L, Khattab AA, Huber C, Gloekler S, Nietlispach F, Mattle H, Jüni P, Wenaweser P. Cerebrovascular accidents complicating transcatheter aortic valve implantation: frequency, timing and impact on outcomes. EuroIntervention 2012;8:62-70.
32. Nombela-Franco L, Webb JG, de Jaegere PP, Toggweiler S, Nuis RJ, Dager AE, Amat-Santos IJ, Cheung A, Ye J, Binder RK, van der Boon RM, Van Mieghem N, Benitez LM, Pérez S, Lopez J, San Roman JA, Doyle D, Delarochellière R, Urena M, Leipsic J, Dumont E, Rodés-Cabau J. Timing, predictive factors, and prognostic value of cerebrovascular events in a large cohort of patients undergoing transcatheter aortic valve implantation. Circulation 2012;126:3041-53.
33. Van Mieghem NM, Schipper ME, Ladich E, Faqiri E, van der Boon R, Randjgari A, Schultz C, Moelker A, van Geuns RJ, Otsuka F, Serruys PW, Virmani R, de Jaegere PP. Histopathology of embolic debris captured during transcatheter aortic valve replacement. Circulation 2013;128:e478-9.
34. Grube E, Naber C, Abizaid A, Sousa E, Mendiz O, Lemos P, Kalil Filho R, Mangione J, Buellesfeld L. Feasibility of transcatheter aortic valve implantation without balloon pre-dilation: a pilot study. JACC Cardiovasc Interv 2011;4:751-7.
35. Rezq A, Basavarajaiah S, Latib A, Takagi K, Hasegawa T, Figini F, Cioni M, Franco A, Montorfano M, Chieffo A, Maisano F, Corvaja N, Alfieri O, Colombo A. Incidence, management, and outcomes of cardiac tamponade during transcatheter aortic valve implantation: a single-center study. JACC Cardiovasc Interv 2012;5:1264-72.
36. Piazza N, Nuis RJ, Tzikas A, Otten A, Onuma Y, García-García H, Schultz C, van Domburg R, van Es GA, van Geuns R, de Jaegere P, Serruys PW. Persistent conduction abnormalities and requirements for pacemaking six months after transcatheter aortic valve implantation. EuroIntervention 2010;6:475-84.
37. Roten L, Wenaweser P, Delacrétaz E, Hellige G, Stortecky S, Tanner H, Pilgrim T, Kadner A, Eberle B, Zwahlen M, Carrel T, Meier B, Windecker S. Incidence and predictors of atrioventricular conduction impairment after transcatheter aortic valve implantation. Am J Cardiol 2010;106:1473-80.
38. Bates MG, Matthews IG, Fazal IA, Turley AJ. Postoperative permanent pacemaker implantation in patients undergoing trans-catheter aortic valve implantation: what is the incidence and are there any predicting factors? Interact Cardiovasc Thorac Surg 2011;12:243-53.
39. Nazif TM, Dizon JM, Hahn RT, Xu K, Babaliaros V, Douglas PS, El-Chami MF, Herrmann HC, Mack M, Makkar RR, Miller DC, Pichard A, Tuzcu EM, Szeto WY, Webb JG, Moses JW, Smith CR, Williams MR, Leon MB, Kodali SK, PARTNER Publications Office PART. Predictors and clinical outcomes of permanent pacemaker implantation after transcatheter aortic valve replacement: the PARTNER (Placement of AoRticTraNscathetER Valves) trial and registry. JACC Cardiovasc Interv 2015;8:60-9.
40. Nuis RJ, Van Mieghem NM, Schultz CJ, Tzikas A, Van der Boon RM, Maugenest AM, Cheng J, Piazza N, van Domburg RT, Serruys PW, de Jaegere PP. Timing and potential mechanisms of new conduction abnormalities during the implantation of the Medtronic CoreValve System in patients with aortic stenosis. Eur Heart J 2011;32:2067-74.
41. Colombo A, Latib A. Left bundle branch block after transcatheter aortic valve implantation: inconsequential or a clinically important endpoint? J Am Coll Cardiol 2012;60:1753-5.
42. Takagi H, Niwa M, Mizuno Y, Goto SN, Umemoto T; All-Literature Investigation of Cardiovascular Evidence Group. Incidence, predictors, and prognosis of acute kidney injury after transcatheter aortic valve implantation: a summary of contemporary studias using Valve Academic Research Consortium definations. Int J Cardiol 2013;168:1631-5.
Cite This Article
Export citation file: BibTeX | RIS
OAE Style
Depboylu BC, Yazman S, Harmandar B. Complications of transcatheter aortic valve replacement and rescue attempts. Vessel Plus 2018;2:26. http://dx.doi.org/10.20517/2574-1209.2018.39
AMA Style
Depboylu BC, Yazman S, Harmandar B. Complications of transcatheter aortic valve replacement and rescue attempts. Vessel Plus. 2018; 2: 26. http://dx.doi.org/10.20517/2574-1209.2018.39
Chicago/Turabian Style
Depboylu, Burak Can, Serkan Yazman, Bugra Harmandar. 2018. "Complications of transcatheter aortic valve replacement and rescue attempts" Vessel Plus. 2: 26. http://dx.doi.org/10.20517/2574-1209.2018.39
ACS Style
Depboylu, BC.; Yazman S.; Harmandar B. Complications of transcatheter aortic valve replacement and rescue attempts. Vessel Plus. 2018, 2, 26. http://dx.doi.org/10.20517/2574-1209.2018.39
About This Article
Special Issue
Copyright
Data & Comments
Data
 Cite This Article 90 clicks
Cite This Article 90 clicks













Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at support@oaepublish.com.